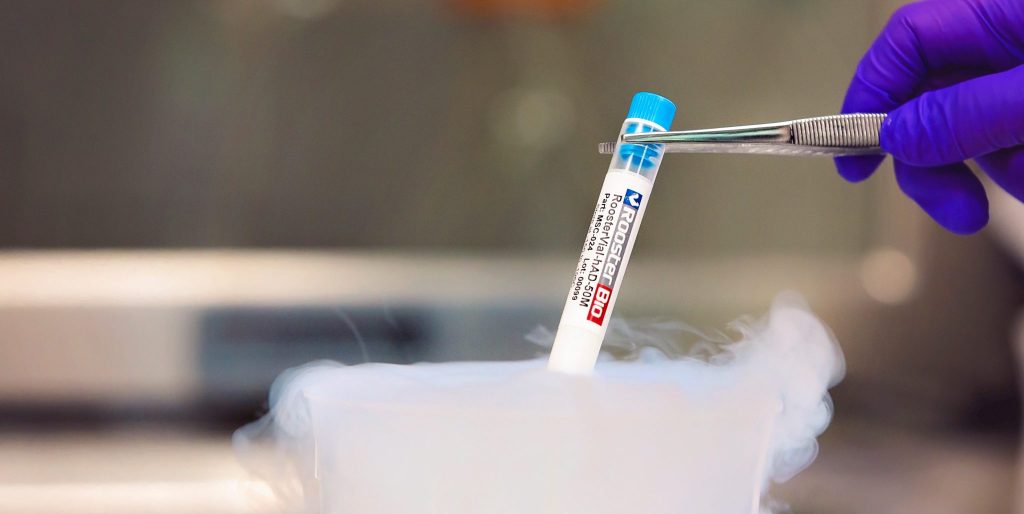
RoosterBio has announced the launch of the Regenerative Medicine (RM) industry’s first xeno-free (XF) hMSC cell banks to advance and simplify stem cell biomanufacturing. By incorporating XF starting materials at early stages, RM scientists can perform more clinically relevant work, resulting in an expedited path towards clinical testing programs. Due of the importance of this announcement, I followed up with Dr. Priya Baraniak, Director of BD & Strategic Marketing at RoosterBio, to further explore details of the launch. [Read more…]
RoosterBio has announced the launch of the Regenerative Medicine (RM) industry’s first xeno-free (XF) hMSC cell banks to advance and simplify stem cell biomanufacturing. By incorporating XF starting materials at early stages, RM scientists can perform more clinically relevant work, resulting in an expedited path towards clinical testing programs. Due of the importance of this announcement, I followed up with Dr. Priya Baraniak, Director of BD & Strategic Marketing at RoosterBio, to further explore details of the launch. [Read more…]
Cynata Therapeutics Initiates Patient Treatment in World 1st Clinical Trial
In breaking news released earlier this month, Cynata Therapeutics (ASX: CYP) announced a major world first. The company treated a patient with steroid resistant acute graft-versus-host disease (GvHD) as part of a phase 1 clinical study of its first mesenchymal stem cell (MSC) product, CYP-001 .
This is an important value catalyst for Cynata and also for the regenerative medicine industry, because it is the first time ever that a patient has been treated with an allogeneic induced pluripotent stem cell (iPSC)-derived therapeutic mesenchymal stem cell (MSC) product.
Webinar: hiPSC-based disease modeling & drug screening for neurodegenerative disorders
 Human induced pluripotent stem cell (iPSC) technology offers the benefits of a cell line, coupled with the advantage of using human primary cells. Applied StemCell (ASC) has developed a panel of iPSC lines for neurotoxicity assays and disease modeling applications. [Read more…]
Human induced pluripotent stem cell (iPSC) technology offers the benefits of a cell line, coupled with the advantage of using human primary cells. Applied StemCell (ASC) has developed a panel of iPSC lines for neurotoxicity assays and disease modeling applications. [Read more…]
Patient Treatment Commences in Cynata’s World First Clinical Trial (CYP-001)
- First patient with steroid resistant acute graft-versus-host disease (GvHD) treated in Cynata’s phase 1 clinical study of its mesenchymal stem cell (MSC) product, CYP-001
- World first clinical trial using a MSC therapeutic product derived from an induced pluripotent stem cell sourced from a single blood donation from one donor
- Transition into active clinical trials cements Cynata’s world leading position in the development and commercialisation of second generation stem cell products
- Partnership with Fujifilm for the commercialisation of CYP-001; trial commencement accelerates this target
How to find unique market opportunities by stem cell type
As the first and only market research firm to specialize in the stem cell industry, BioInformant’s 175-page global strategic report “Stem Cell Research Products – Opportunities, Tools, and Technologies” evaluates strategies across the stem cell product spectrum, with emphasis on opportunities for research product development.
It explores unique market opportunities by stem cell type, including mesenchymal stem cells (MSCs), hematopoietic stem cells (HSCs), induced pluripotent stem cells (iPSCs), embryonic stem cells (ESCs), neural stem cells (NSCs), and more. [Read more…]
- « Previous Page
- 1
- …
- 105
- 106
- 107
- 108
- 109
- …
- 194
- Next Page »