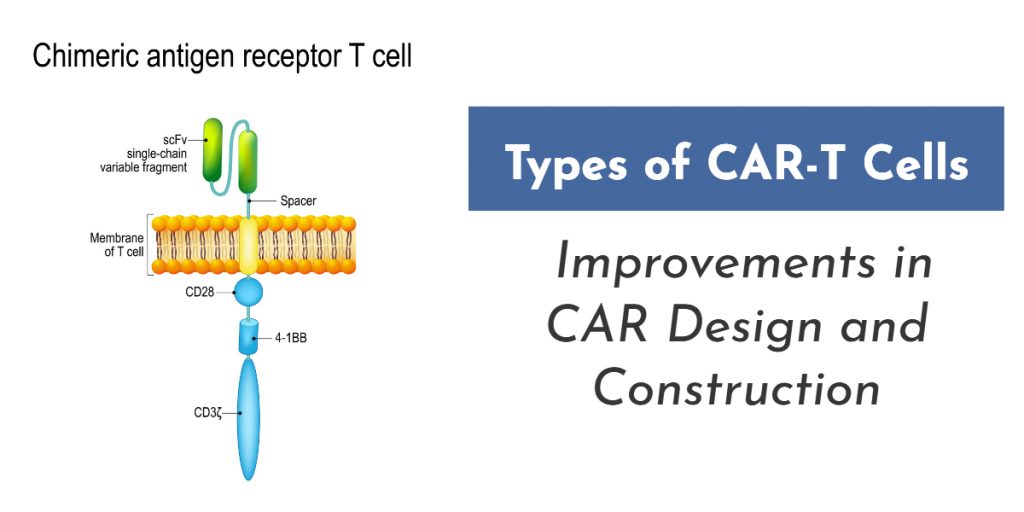
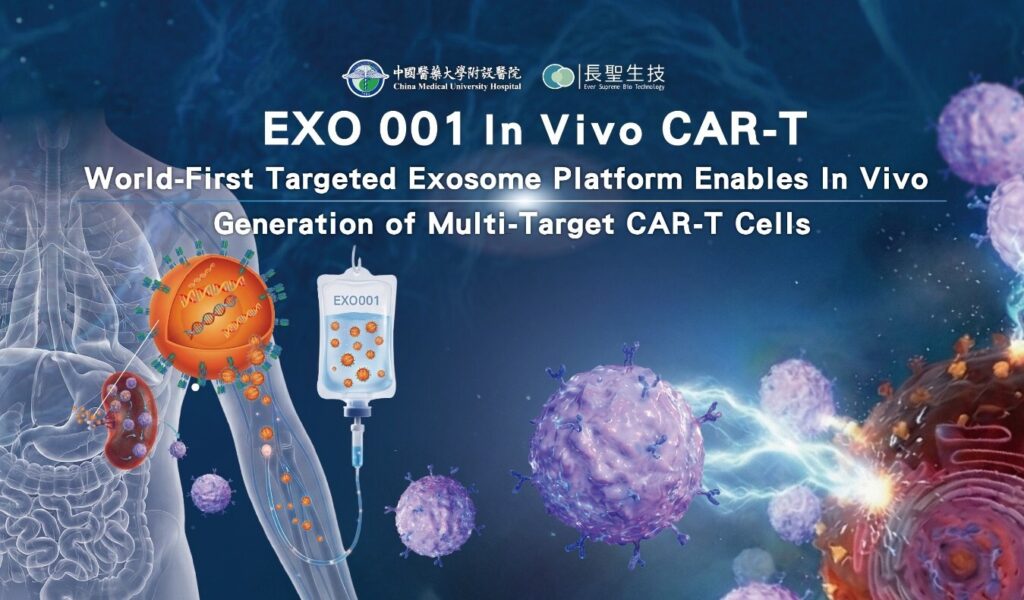
What are the types of CAR-T cells and how have they changed in recent years? CAR-T cells are immune cells that are artificially engineered to express a particular chimeric antigen receptor (CAR), which have shown great potential in killing cancer cells. Chimeric antigen receptors (CARs) are artificially produced recombinant receptors having both antigen-binding and T-cell activating potentials.
CARs are composed of an extracellular recognition domain sourced from monoclonal antibodies (mAbs) and an intracellular signaling domain including CD3- and costimulatory domains for triggering endogenous signaling pathways of T-cells. [Read more…]