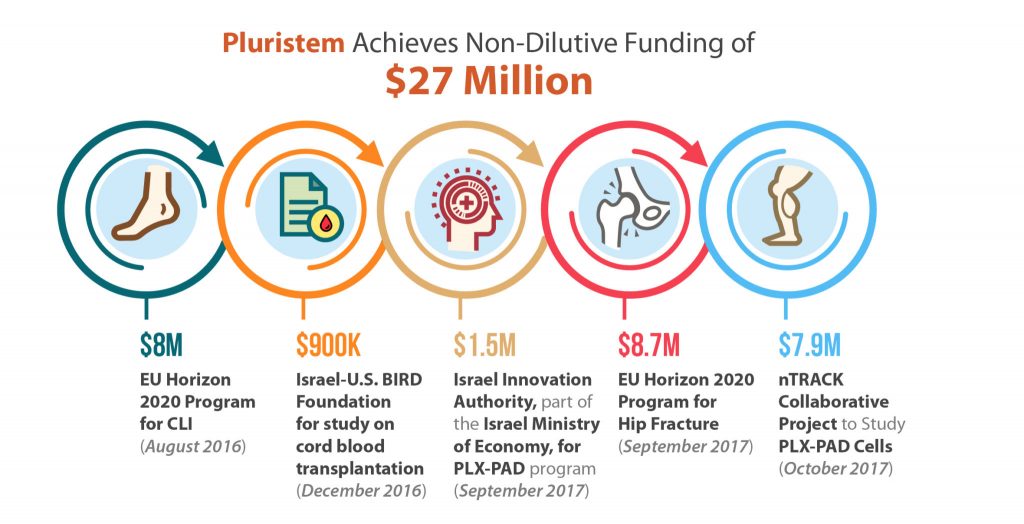
Non-dilutive funding, in substantial amounts, is something nearly all biotechnology companies want, and few get. Although small grants from various government agencies like the NIH are helpful, and are available to a larger number of biotechs, Pluristem Therapeutics (Nasdaq:PSTI) has accomplished something quite unique in the last year as the recipient, of three awards totaling nearly $25 million from the European Union’s Horizon 2020 program as well as $2.4 million in smaller grants from Israel. A majority of these funds are aimed at bringing Pluristem’s Phase III assets to market and to better predict outcomes of cell therapy treatments.
Pluristem’s lead asset, its PLX-PAD cell therapy, is currently in a multinational pivotal Phase III study for the treatment of critical limb ischemia (CLI) in 250 patients with CLI Rutherford Category 5 who are unsuitable for revascularization. These patients have very few remaining treatments options and will most likely be headed for amputation. Cell therapy may be the answer. The U.S. FDA has given Pluristem Fast Track Designation in this indication, while the European Medicines Agency (EMA) has selected the PLX-PAD program in CLI for its Adaptive Pathways project, the goal of which is to shorten time to market for innovative medicines. Europe gave the PLX-PAD program in CLI one more boost with the award of $8 million last August from the EU’s Horizon 2020 program, Europe’s largest research and innovation program. Clearly, the Europeans are eager for a novel cell therapy to treat CLI and they are banking on Pluristem to bring it to market quickly. [Read more…]