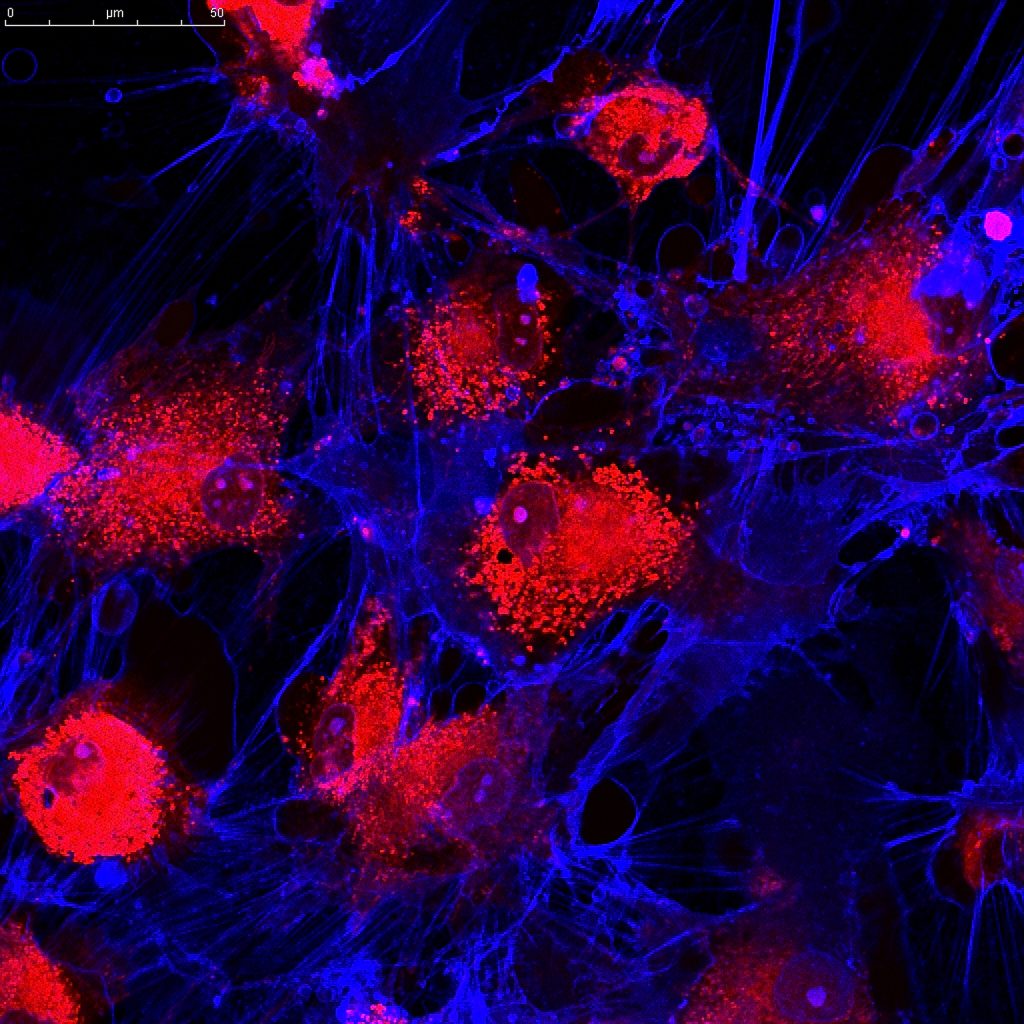
Currently over 50% of men and women are directed to unnecessary treatment or surgery. A new approach may help. Professor Alan Doucette of Dalhousie University and his colleagues have published a study funded by the Breast Cancer Society of Canada in which they capture particles released by rapidly growing cells called exosomes. Like parcels containing tools and instructions, exosomes contain an “inventory of intent.” Meaning, the cells that have released them contain genetic material proteins and metabolites that promote invasion of normal cells and commandeer their resources. [Read more…]
Currently over 50% of men and women are directed to unnecessary treatment or surgery. A new approach may help. Professor Alan Doucette of Dalhousie University and his colleagues have published a study funded by the Breast Cancer Society of Canada in which they capture particles released by rapidly growing cells called exosomes. Like parcels containing tools and instructions, exosomes contain an “inventory of intent.” Meaning, the cells that have released them contain genetic material proteins and metabolites that promote invasion of normal cells and commandeer their resources. [Read more…]
Commence Bio Receives 1st Patent for MSC1, An Innovative Cancer Immunotherapy Platform
In major news released this week, Commence Bio announced that the company has received its first patent for MSC1. Commence Bio is a pre-clinical stage biotechnology company that is based on the concept that cancer and other inflammatory conditions can be addressed by “rebooting patients’ immune systems” with a new class of medicinal stem cells, MSC1 and MSC2.
The company is now developing a therapeutic pipeline, as well as offering research products that include MSC1 and MSC2 cells and media. The concept around which the company is based is that its proprietary “stimulated Toll-like receptor technology” programs naïve MSCs to either a more potent anti-inflammatory phenotype, or a new pro-inflammatory, anti-tumor phenotype. [Read more…]
Commence Bio Focuses on Mesenchymal Stem Cells (MSCs) as Immunotherapies with New Website and Corporate ID
Commence Bio is an intriguing company that has recently entered the mesenchymal stem cell (MSC) marketplace. Commence Bio came across my radar screen earlier this year right around the time that it announced the name change, as highlighted in the company press release below.
Cynata Partners with Harvard University and University of Sydney
In a recent “Activity Report” released by Cynata Therapeutics, the company announced the status of key business activities and partnerships, including a recent collaboration with Harvard Medical School to treat cancer using toxin-secreting stem cells. It has also entered into a collaboration with the University of Sydney that will address myocardial infarction using the therapeutic properties Cynata’s iPSC-derived mesenchymal stem cells (MSCs).
In addition, the company recently announced that their Cymerus™ derived MSCs will be utilized in a clinical trial for GvHD, the first in-patient test of the company’s novel stem cell manufacturing process.
These activities are resulting from Cynata’s proprietary manufacturing platform that can produce unlimited quantities of mesenchymal stem cells (MSCs) by deriving them from induced pluripotent stem cells (iPSCs) produced from a single donor. This Cymerus™ technology has made Cynata one of the first commercial providers to achieve consistent, scalable, and importantly, cost-effective production of clinical-grade stem cells. The company’s novel manufacturing approach is overcoming a major hurdle that has traditionally limited the therapeutic use of stem cells, which is the “sky high costs” associated with such procedures. [Read more…]