Cord blood market segmentation is one of the most important (and least appreciated) parts of analyzing the cord blood banking industry. As the first and only market research firm to track the stem cell industry, BioInformant has collected over 16 years of historical information on the cord blood industry from 2006 to present. This provides a powerful record if past industry dynamics, as well as a robust platform for making future projections. [Read more…]
Mesenchymal Stem Cells (MSCs)
Mesenchymal stem cells (MSCs) are a well-characterized population of adult stem cells that can differentiate into a variety of cell types (chondrocytes, osteoblasts, adipocytes, myocytes, and more).
denovoMATRIX Launches Novel Microcarrier to Advance Cell and Gene Therapy Manufacturing
DRESDEN, Germany, May. 04, 2022 — denovoMATRIX GmbH, an innovator in cell and gene therapy (CGT) manufacturing, announced the launch of a novel microcarrier for increasing access to high-quality mesenchymal stromal cells (MSCs) for stem cell-based therapies. The product, termed beadMATRIX, is designed to enable MSC proliferation and harvest in xeno-/serum-free media with high efficiency and stemness.
Forever Labs, Is It Worth $7,000 to Save Cells from Your Younger Self?
Forever Labs is one of the first companies worldwide to offer stem cell storage for adults. Interestingly, there is a prolific market for the storage of newborn tissues, including umbilical cord blood, umbilical tissue, and placental blood and tissue. [Read more…]
Cultured Meat Market: From the Lab to the Grocery Store
Cultured meat refers to lab-grown meat created using cell culture techniques. It is produced by growing master cells collected from cattle, chicken, pigs, fish, and lamb, as well as other types of livestock and seafood. In addition to meat and seafood production, cultured meat techniques can be used to ethically manufacture other types of animal products, such as leather, fur, milk, and hen-free egg whites, for example. Cultured meat is ethically produced, because livestock is not used within the manufacturing processes beyond collecting the initial cells for culture. [Read more…]
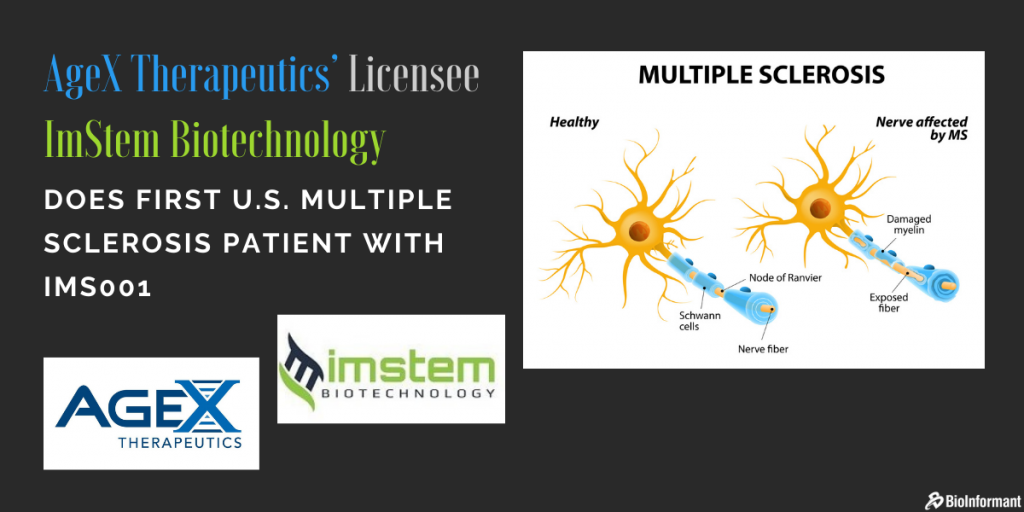
AgeX Therapeutics’ Licensee ImStem Biotechnology Does First U.S. Multiple Sclerosis Patient with IMS001
-
ImStem’s MSC Product Candidate IMS001 is Derived from AgeX’s Pluripotent Stem Cell Line ESI-053.
-
IMS001 is an investigational, allogeneic, MSC product derived from a human embryonic stem cell line.
-
It is assumed to be the first hES-MSC based allogeneic cell therapy accepted for clinical trial by the FDA for patients with MS.
- « Previous Page
- 1
- …
- 6
- 7
- 8
- 9
- 10
- …
- 61
- Next Page »