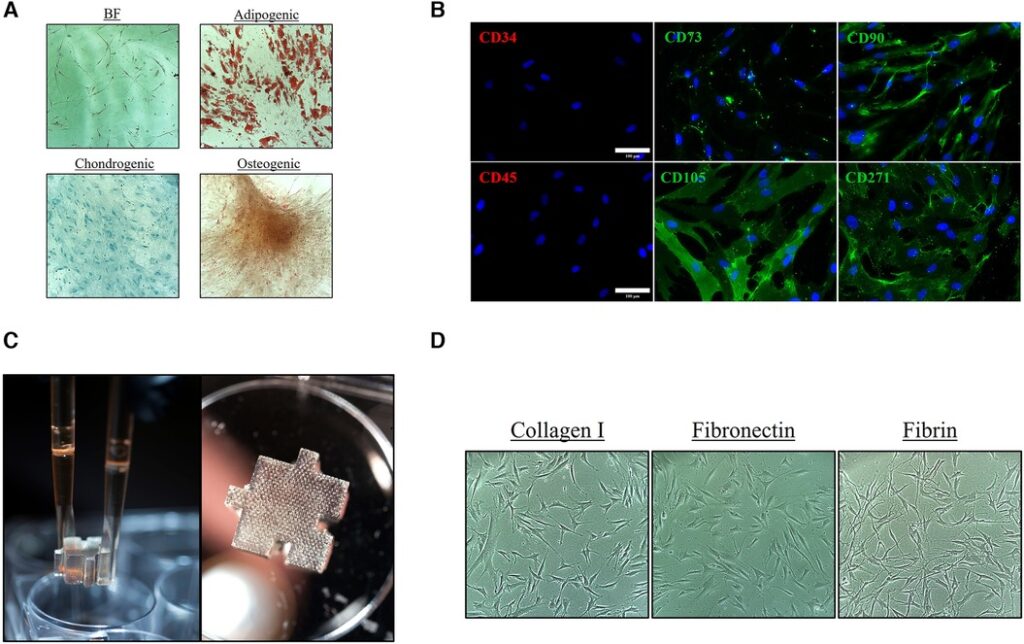
Overland Park, KS – A groundbreaking study titled “Tailoring the Secretome Composition of Mesenchymal Stem Cells to Augment Specific Functions of Epidermal Regeneration: An In Vitro Diabetic Model,” conducted by, Jacob G. Hodge, Ph.D., introduces a pioneering strategy to revolutionize epidermal regeneration using the secretome of mesenchymal stem cells (MSCs) utilizing Ronawk’s Bio-Blocks™. Published in June 2023 in the journal
“Frontiers in Medical Technology” and available through open access, the study unveils a powerful solution for optimizing wound healing, particularly in a diabetic context. [Read more…]
Mesenchymal Stem Cells (MSCs)
Mesenchymal stem cells (MSCs) are a well-characterized population of adult stem cells that can differentiate into a variety of cell types (chondrocytes, osteoblasts, adipocytes, myocytes, and more).
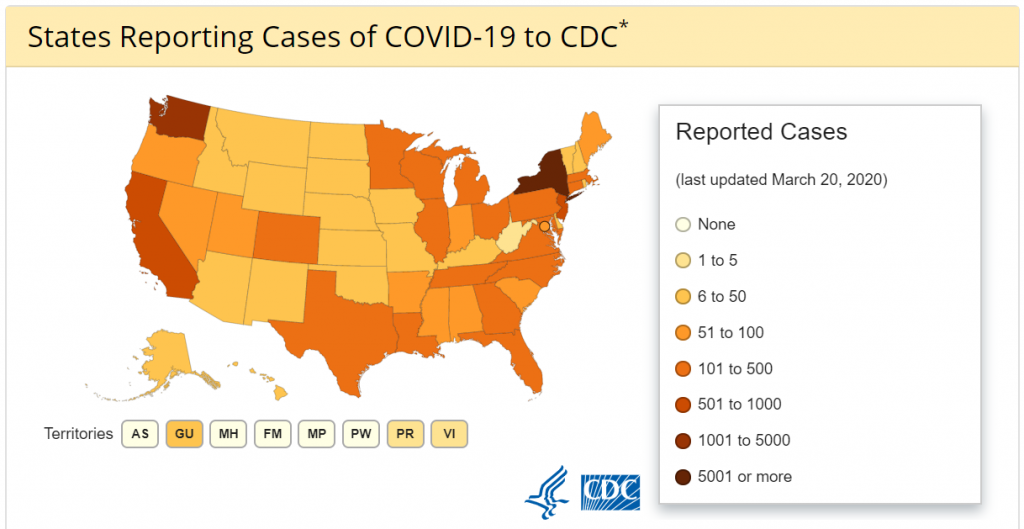
Could Cynata’s Cymerus™ Platform Be A Tool in the War Against Coronavirus?
What is the Cymerus™ technology and how could it potentially assist in the battle against Coronavirus cases on a global basis? Cymerus™ is a stem cell manufacturing platform being developed by Cynata Therapeutics Limited (ASX: CYP), an Australian regenerative medicine company.
The proprietary technology utilizes induced pluripotent stem cells (iPSCs) originating from an adult donor as the starting material for generating mesenchymoangioblasts (MCAs). It then differentiates these cells into mesenchymal stem cells (MSCs). [Read more…]
PBS Biotech and denovoMATRIX Join Forces to Boost Manufacturing Processes for Cell & Gene Therapies
Camarillo, CA (April 11, 2023) – PBS Biotech, an innovative, single-use bioreactor manufacturer and process development services provider, announces its collaboration with denovoMATRIX, a leading supplier of next-gen biomimetic coating technologies. By joining forces, PBS Biotech and denovoMATRIX seek to foster xeno-free expansion and scale-up of clinical-grade mesenchymal stromal cells (MSCs) and induced pluripotent stem cells (iPSCs) for use in allogeneic and autologous cell and gene therapies. The first outcome of this collaboration is a white paper presenting the advantages in MSC manufacturing that can be achieved using their cutting-edge technologies. [Read more…]
FDA Grants RMAT Designation for Rexlemestrocel-L in Chronic Low Back Pain
NEW YORK, Feb. 08, 2023 — Mesoblast Limited (Nasdaq:MESO; ASX:MSB), global leader in allogeneic cellular medicines for inflammatory diseases, today announced that the United States Food and Drug Administration’s (FDA) Office of Tissues and Advanced Therapies (OTAT) has granted Regenerative Medicine Advanced Therapy (RMAT) designation for rexlemestrocel-L in the treatment of chronic low back pain (CLBP) associated with disc degeneration, in combination with hyaluronic acid (HA) as delivery agent for injection into the lumbar disc. [Read more…]
The Future Forecast for MSC-Based Therapeutics
Presently, MSC-based clinical trials are being conducted for a range of disease conditions, with many completed trials showing safety and efficacy. Clinical utility of MSCs is primarily attributed to their four key biological properties, which include: [Read more…]
- « Previous Page
- 1
- …
- 3
- 4
- 5
- 6
- 7
- …
- 61
- Next Page »