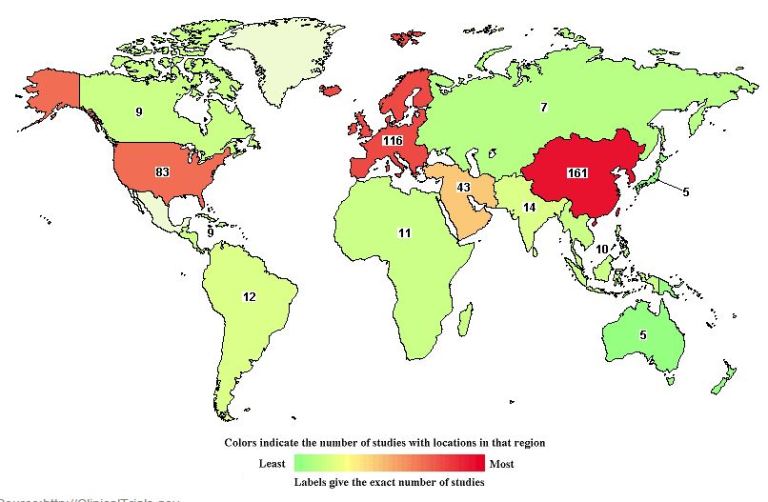
Historical Number of Mesenchymal Stem Cell Clinical Trials (IFCB)
In 2014, the International Federation for Cell Biology (IFCB) conducted an analysis of mesenchymal stem cell clinical trials, identifying that 313 clinical trials involved mesenchymal stem cells (MSCs). The majority of these clinical trials (41.7%) were in Phase I/II. Another 23.7% of these studies were in Phase II. However, only 4.2% of these clinical trials had progressed to Phase III. As such, it is clear that the bulk of MSC clinical trials remain in the early stages of development.
Among the clinical trials that progressed through Phase III, one of the most promising stem cell therapies to emerge was Prochymal, an allogenic mesenchymal stem cell therapy derived from the bone marrow of adult donors and made by Osiris Therapeutics. Prochymal is produced by purifying MSCs from the bone marrow of donors, then culturing and packaging the cells, allowing up to 10,000 doses to be derived from a single donor. [Read more…]