CryoHoldco is a stem cell bank holding company that is the market leader in Latin America and one of the top 10 largest cord blood banks in the world. CryoHoldco is now seven times larger than any other stem cell bank in Latin America with over 275,000 stem cell units in storage.
Cord Blood Banking in Latin America – Interview with James Mendez of Cryoholdco
In this interview with James Alexis Mendez, we discuss the market strategy of CryoHoldco, a stem cell bank holding company that is the market leader in Latin America and one of the ten largest cord blood banks in the world. James Alexis Mendez currently serves as the CFO and Board member of CryoHoldco. Approaching 200,000 stem cell units in storage, CryoHoldco is more than 5X larger than any other stem cell bank in Latin America. CryoHoldco owns the cord blood market leaders in both Mexico and Colombia, and plans to expand into other regions of Latin America. [Read more…]
Stem Cell Thought-Leaders—and What You Can Learn From Them
Without question, some of BioInformant’s most popular content has been our interviews with key opinion leaders (KOL). Highlighted below are five interviews that have been widely shared across the stem cell industry. The first interview with Dr. Sadiq has gotten more than 2,200 shares on social media, so you definitely need to check it out. Enjoy!
Stem Cell Therapy Shows Promise for Treatment of MS – Q&A with Dr. Sadiq of the Tisch MS Research Center of NY
In breaking news from the Tisch MS Research Center of New York, Dr. Sadiq announced he is pursuing a Phase II clinical trial exploring the use of stem cells in the treatment of MS. The announcement followed positive results from a FDA-approved Phase I stem cell trial in which MS patients received multiple spinal injections of neural progenitors derived from bone marrow mesenchymal stem cells (called MSC-NPs). Compellingly, it was the first time ever that a treatment method has demonstrated reversal of established disability in MS patients. [Read more…]
Comparison of the Top 3 International Cord Blood Banks – Cryo-Save, CryoHoldco, CCBC
In a recent article, BioInformant compared the top three largest cord blood banks within the United States. The purpose of the analysis was to consider comparative metrics between the largest market leaders in the cord blood industry within the United States, the most mature cord blood market worldwide.
In this article:
- How to Choose a Cord Blood Bank
- Top 3 International Cord Blood Banks
- Comparison of Top 3 International Cord Blood Banks
The Best International Cord Blood Banks
In this article, we identify three of the best cord blood banks in the world and explore what makes them the market leaders.
How to Choose a Cord Blood Bank?
In this article, we compare the three of the leading cord blood banks outside of the United States. The metrics used to identify the top international cord blood banks were: 1) Number of cord blood and cord tissue units stored, 2) Number of units released for transplant, 3) Rate of growth, and 4) Geographic reach (regions served).
The three companies identified as the market leaders in this international analysis are: Cryo-Save, CryoHoldco, and China Cord Blood Corporation (CCBC).
Top 3 International Cord Blood Banks
1. Cryo-Save | Previously Cryo-Cell Cord Blood Bank
Located in Zutphen, Netherlands, Cryo-Save is the largest cord blood bank in Europe, with more than 268,000 cord blood and tissue stem cell units in its storage capacity.1 Cyro-Save was founded in 2000 and changed its name from Cyro-Cell to Cyro-Save in 2003. Its subsidiary, Cryo-Save Arabia, was the first private cord blood bank to initiate operations in the Middle East when it launched on September 11, 2006. Cryo-Save has stored stem cell samples from over 70 countries on six continents, using processing and storage facilities in Belgium, Germany, Dubai, India, and South Africa.
The Sepax System Processing Method
Cryo-Save uses an automated processing method, the Sepax system, developed by the Swiss company Biosafe. The Sepax system is a fully-automated system for separating cellular components: a light beam identifies the density gradient between various cell layers after cellular separation has been achieved, by spinning down the full blood sample within a centrifuge. It uses a bag system that is a single-use, sterile, fully-closed system.
International Influence in Cord Blood Banking
The company uses its “Cryo-Save” trademark across more than 40 countries, where company divisions and business partners operate under licensing agreements. Cord blood and cord tissue units from most operating divisions and licensees are stored in their central Belgium facility, with the exception of operators functioning in Germany, Dubai, India, and South Africa. Its primary Belgium facility has both AABB accreditation and ISO 9001:2008 certification. Its other laboratories are well credentialed, too.
Diverse Stem Cell Biobanking
Interestingly, similar to several other operators within Europe (most notably within the UK), Cryo-Save is following the emerging trend of diversifying into additional types of stem cell biobanking for purposes of augmenting its central cord blood banking service. The company now offers cord tissue banking services across all of its branches. In May 2010, the company also launched an adipose tissue stem cell storage service, called “Cryo-Lip.”2
Cryo-Save Storage Services
The company’s three stem cell storage services are shown below:
- Hematopoietic stem cells obtained from the umbilical cord blood (“Cryocord”)
- Mesenchymal stem cells obtained from the umbilical cord tissue (“Cryocord+”)
- Mesenchymal stem cells obtained from adipose tissue (“Cryo-Lip)
Cryo-Save Global Labs
Cryo-Save has multiple labs located across the globe, with a strong presence in Europe, as shown below:
- Europe: Niel/Antwerp, Belgium (2009); Aachen, Germany (2009); Lyon, France (2014)
- Rest-of-World: Dubai, UAE (2006); Bangalore, India (2008); Cape Town, South Africa (2011)
2. CryoHoldco | Acquiring Cord Blood Banking Companies
CryoHoldco is a stem cell bank holding company that is the market leader in Latin America and one of the largest stem cell banks in the world. CryoHoldco is about five times larger (5X) than any other stem cell bank in Latin America—with more than 125,000 cord blood samples stored. When cord tissue units are also included, it has more than 130,000 stem cell units in inventory.
Advantages of Controlling Cryo-Cell Mexio as an Asset
CryoHoldco was created in 2015 with the purpose of forming the premier stem cell banking company in Latin America. The first asset that was acquired by CryoHoldco was Cryo-Cell Mexico. Cryo-Cell Mexico (CCM) was founded in 2002 by local entrepreneurs in Guadalajara, Mexico, after an investigation of stem cell therapies that was prompted by the illness of a family member.
CCM was the first stem cell bank in Latin America. Since investing in Cryo-Cell Mexico in mid 2015, CryoHoldco has been active in the market and has acquired a controlling interest in a regional stem cell bank in Mexico (Q4 of 2015) and has created a joint venture with a smaller bank that is affiliated and has exclusive access to the leading chain of private hospitals in Mexico (Q1 2016).
Acquisition of Stem Medicina Regenerativa
Notwithstanding its activity in Mexico, CryoHoldco’s scope and focus is pan-regional. This led it to the acquisition of Stem Medicina Regenerativa, the largest stem cell bank in Colombia, in early 2016.
The Largest Cord Blood Bank in Latin America
Today, CryoHoldco represents the largest stem cell banking operation in Latin America, with over 130,000 stored samples, two state-of-the-art lab and storage facilities in Mexico and Colombia, a growing network of 10,000+ affiliated doctors, and over 100 sales offices across our countries and regions of operation. CryoHoldco’s state-of-the-art storage facility in Guadalajara, Mexico, has a capacity of 300,000 samples.
Tissue and Cord Blood Banking
Like other leading stem cell banks, CryoHoldco has a diversified revenue stream and product offering, which includes cord blood and cord tissue banking, as well as storage of mesenchymal cells derived from dental pulp. In 2016 the company will also launch adipose tissue storage and a maternity bank.
3. China Cord Blood Corporation
Headquartered in Beijing, China, China Cord Blood Corporation (CCBC) was the first umbilical cord blood bank to begin operating within China.
Private and Public Cord Blood Banking
Recently, the Chinese government established that it would only allow one cord blood bank in each of the Chinese provinces. The Chinese government issues one cord blood banking license per province, and the licensed company must, therefore, function as both a public (donation-based) and private (for-profit) bank. The for-profit division funds the company’s donation-based public division, as no government support is received for this purpose.
The Largest in China
China Cord Blood Corporation currently holds the exclusive license for three of the regions and has partial ownership in a 4th province. All of the other companies hold only one license each. As such, it is also the largest cord blood bank in China, having either exclusive or partial operating rights within regions that account for nearly three-quarters of the annual births within the country. It is also the fastest growing cord blood banks worldwide, according to Mr. Chen Bing Chuen (“Albert”), Chief Financial Officer and Director of the China Cord Blood Corporation.3
CCBC Services
The company’s prime services include:
- cord blood collection
- laboratory testing
- hematopoietic stem cell processing
- stem cell storage services
More About the Corporation
- China Cord Blood Corporation is one of the founders of the highly recognized non-profit organization – AsiaCORD (Asia Cord Blood Bank) Organization.
- The company operates laboratories in Beijing and Guangdong.
- The cord blood bank in Beijing is operated by Beijing Jiachenhong Biological Technologies Co. Ltd., which received its cord blood banking license in September 2002. The Beijing cord blood laboratory was awarded AABB accreditation in early 2013.
- The cord blood bank in Guangdong is operated by Shenzhen Nuoya Pharmaceutical Equipment Co. Ltd, which received its cord blood banking license in June 2006.
Comparison of Top 3 International Cord Blood Banks
A comparative analysis of services and features, as well as pricing, is shown below for these global market leaders.
TABLE. Comparison of Services & Features for Top 3 International Cord Blood Banks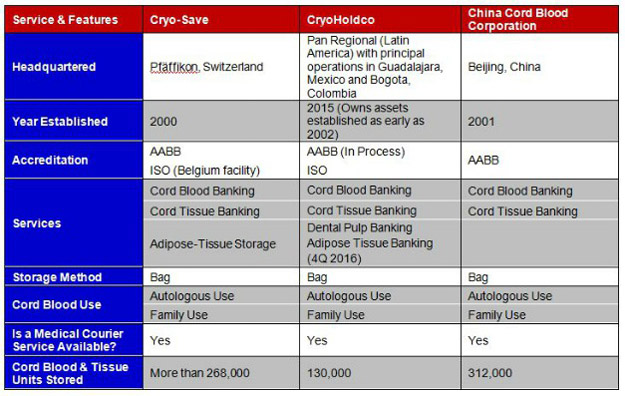
If you found this blog valuable, subscribe to BioInformant’s stem cell industry updates.
As the first and only market research firm to specialize in the stem cell industry, BioInformant research is cited by The Wall Street Journal, Xconomy, AABB, and Vogue Magazine. Bringing you breaking news on an ongoing basis, we encourage you to join more than half a million loyal readers, including physicians, scientists, executives, and investors.
What do you think of these top 3 international cord blood banking companies? Share your thoughts in the comments section below.
Up Next: Cord Blood vs. Cord Tissue Stem Cells
1 Esperite.com, (2014). Cryo-Save news, annual reports and interim reports | www.cryo-save.com. [online] Available at: http://esperite.com/u/news__reports.html?mnSelY=2010&mnSelM=&mnSeliG=62&mnSeliM=634 [Accessed 4 Nov. 2015].
2 Esperite.com, (2014). Cryo-Save news, annual reports and interim reports | www.cryo-save.com. [online] Available at: http://esperite.com/u/news__reports.html?mnSelY=2010&mnSelM=&mnSeliG=62&mnSeliM=634 [Accessed 4 Nov. 2015].
3 Chen, A. (2014). Presentation: The Commercialization of Umbilical Cord Blood Banking in China and its Operational Challenges. World Cord Blood Congress, September 17, 2014.
Editor’s Note: This article was originally published on October 8, 2018, and has been updated for quality and relevancy.
CryoHoldco Acquires Criocord, a Peruvian Cord Blood Bank and Cell Therapy Institute
 CryoHoldco has announced the acquisition of Criocord, a cord blood bank and cell therapy institute in Perù, for an undisclosed sum. CryoHoldco already owns the cord blood market leaders in Mexico and Colombia and now controls an important asset in Peru. A portfolio company of ACON investments, CryoHoldco is a stem cell bank holding company that is the market leader in Latin America and one of the largest cord blood banks worldwide. [Read more…]
CryoHoldco has announced the acquisition of Criocord, a cord blood bank and cell therapy institute in Perù, for an undisclosed sum. CryoHoldco already owns the cord blood market leaders in Mexico and Colombia and now controls an important asset in Peru. A portfolio company of ACON investments, CryoHoldco is a stem cell bank holding company that is the market leader in Latin America and one of the largest cord blood banks worldwide. [Read more…]