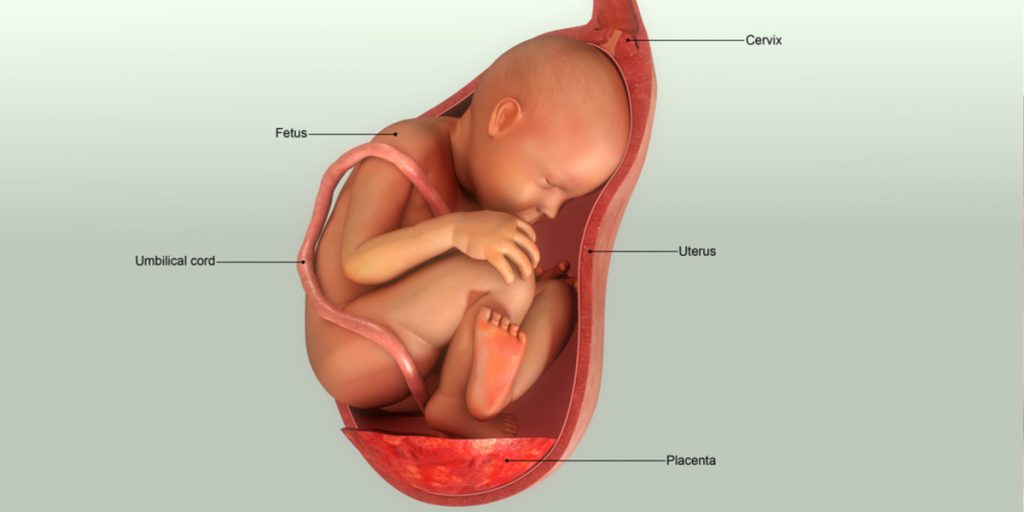
The history of cord blood and tissue banking dates back to the early 1990’s when three private cord blood banks were formed within the United States. These banks were Cord Blood Registry, Cryo-Cell, and Viacord, all of which are AABB accredited banks. Shortly thereafter, cord blood banks began forming in leading healthcare markets worldwide.
In this article: