RoosterBio, Inc, a biotechnology market disrupter and leading supplier of innovative human mesenchymal stem/stromal cell (hMSC) biomanufacturing systems, released the Regenerative Medicine industry’s first human umbilical cord (hUC) MSC paired cell and media system designed for product development and bringing with it an unprecedented step-change in quantifiable productivity metrics. RoosterVial™-hUC-MSC-XF, allows scientists to generate billions of perinatal cells in a fraction of the time when paired with RoosterBio’s existing xeno-free (XF) hMSC plug and play systems. This product line is the only commercially available, high-volume perinatal MSC bioprocess system with a line of sight to towards clinical translation.
“Until now, there was not a hUC-MSC on the market that would support the quality and scale needs of cell therapy product developers,” said RoosterBio Founder & Chief Product Officer Jon A. Rowley, PhD. RoosterBio’s launch of RoosterVial-hUC-MSC-XF introduces another first-in-class hMSC product on the market that simplifies all aspects of product development. “This very powerful set of biological tools has now been released into the Regenerative Medicine marketplace, introducing a major step-change in MSC productivity,” added Dr. Rowley.
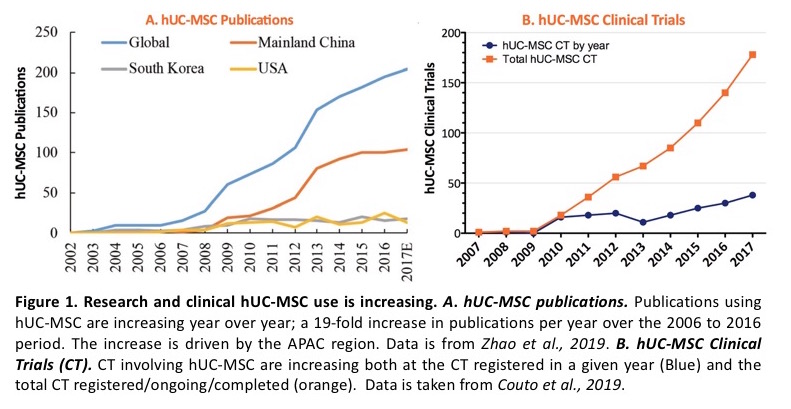
According to recent global publications, the umbilical cord is the fastest growing hMSC tissue source and the second hMSC type in overall usage within hMSC Regenerative Medicine applications. Growth of hUC-MSC use in Asia and Europe has been faster than in North America due to the lack of availability of hUC-MSC critical raw material in North America. “By introducing new tissue types, we are opening the door to support the incorporation of these new living cellular technologies into product development across the globe,” said Dr. Rowley.
This system has several key advantages over the limited number of suppliers of perinatal hMSC. Being XF, and manufactured with RoosterBio’s existing cGMP compatible processes, the system allows for product development and translational research to start with translation-friendly materials. Secondly, by providing these high-quality, critical raw materials at higher cell volumes with increased efficiencies (reduced media consumption and cost per M cell), and right-sized 2D or 3D bioreactor scalable processes – the system allows product developers to focus on their therapeutic applications.
Finally, RoosterBio provides first in class characterization on each donor and manufacturing lot of hMSC key quality attributes (PDL, identity, expansion potential) and functional assays (cytokine secretion, trilineage differentiation, immunomodulation) fostering confidence and additional insight in both experimental reproducibility and robustness of results. Read the RoosterBio blog for more information on these advantages.

The product is backed by licensed intellectual property (IP) from Tissue Regeneration Therapeutics (TRT), a Canadian Controlled Private Corporation with a focus on Human Umbilical Cord PeriVascular Cell (HUCPVC) technology. RoosterBio announced the collaboration with TRT in May 2019 which gave RoosterBio an exclusive worldwide license to TRT’s patent estate related to the manufacture and global sale of human umbilical cord-derived MSCs. The announcement advances both companies’ shared goal of accelerating the development of the Regenerative Medicine industry and gives customers the option for licensing opportunities specific to their therapeutic indication.
Visit https://www.roosterbio.com/collections/high-volume-hmscs/huc-mscs for more information about RoosterBio’s radically simplified xeno-free hUC-MSCs. For a RegMedNet webinar on September 25, 2019 with Dr. Rowley entitled “Rapid Clinical Translation and Scale-Up of hMSCs and EVs”, register here.
About RoosterBio, Inc
RoosterBio, Inc. is a privately held cell manufacturing platform technology company focused on accelerating the development of a sustainable regenerative medicine industry, one customer at a time. RoosterBio’s products are high-volume, affordable, and well-characterized adult human mesenchymal stem/stromal cells (hMSCs) paired with highly engineered media systems. RoosterBio has simplified and standardized how stem cells are purchased, expanded, and used in development, leading to marked time and costs savings for customers. RoosterBio’s innovative products are ushering in a new era of productivity and standardization into the field, accelerating the road to discovery in Regenerative Medicine. For more information on RoosterBio, please visit http://www.roosterbio.com.
Do you have questions for RoosterBio? Ask them in the comments below.


















Congratulations for your new innovation.
Like to hear more about your technology , prices and if you offer training to Medical doctors interested in perinatal cell therapy for Regenerative Medicine.
Thanks.
I am 58:years old with MS considering going to Cayman Islands for stem cell therapy, I do not believe I the medicines and have been on a diet and vitamin D protocol and hoping for relief with stem umbilical therapy. Is this a good choice