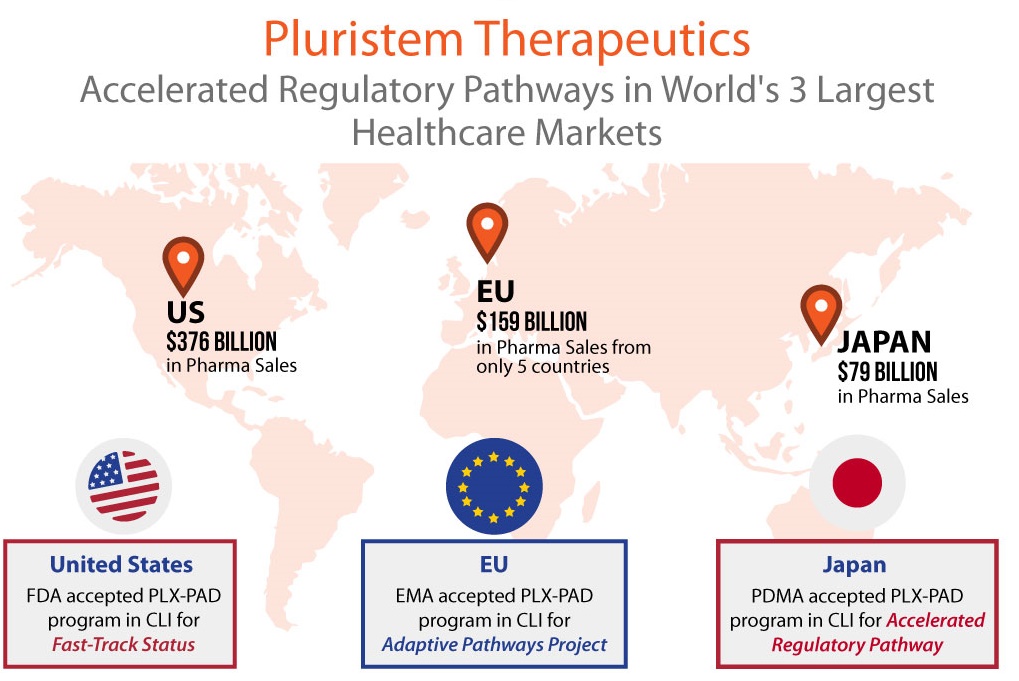
Cell therapy was recently described as “One of the most promising new fields of science and medicine,” by FDA Commissioner Scott Gottlieb. He went on to state that cell therapy and regenerative medicine “…hold significant promise for transformative and potentially curative treatments for some of humanity’s most troubling and intractable maladies.” In the FDA statement issued August 28th, Gottlieb also revealed that the U.S. regulator is looking improve upon its approval process for cell therapies. [Read more…]
Stem Cell News
BioInformant's coverage of stem cell news includes iPS cells, mesenchymal stem cells, hematopoietic stem cells, neural stem cells and more.
As the first and only market research firm to specialize in stem cell news, BioInformant research has been cited by the Wall Street Journal, Xconomy, AABB, and Vogue Magazine.
Serving Fortune 500 leaders that include GE Healthcare, Pfizer, and Goldman Sachs, BioInformant is your global leader in stem cell industry news.
Plasticell and Anthony Nolan to partner in clinical trials of ex vivo expanded cord blood stem cells
 December 5 2017, Stevenage, UK–Plasticell, a developer of innovative stem cell technologies and cell therapies, today announced it will collaborate with Anthony Nolan, a leading research organisation dedicated to saving the lives of people with blood cancers, to progress clinical development of Plasticell’s ex vivo expanded cord-blood derived hematopoietic stem cell product.
December 5 2017, Stevenage, UK–Plasticell, a developer of innovative stem cell technologies and cell therapies, today announced it will collaborate with Anthony Nolan, a leading research organisation dedicated to saving the lives of people with blood cancers, to progress clinical development of Plasticell’s ex vivo expanded cord-blood derived hematopoietic stem cell product.
Allogeneic haematopoietic stem cell transplantation (HSCT) is a curative therapy for patients with life-threating haematological disorders and the most successful cell replacement therapy developed to date with over 40,000 transplantations performed per year worldwide. Cord blood (CB) is an ideal starting point for allogeneic HSCT as it can be collected and banked without compromising the donor and is more easily matched to recipients relative to bone marrow–derived material. However, the limited number of stem cells available in a single cord unit limit their use to paediatric patients and can cause delayed engraftment. [Read more…]
ISCT Webinar on Cell and Gene Therapy (CGT) Products in Japan
Have you heard of the recent regulatory changes surrounding cell and gene therapy (CGT) products in Japan?
These changes have greatly increased the interest in taking CGT clinical programs to Japan from development through commercialization. ISCT (International Society for Cellular Therapy) and FIRM (Forum for Innovative Regenerative Medicine) have created a partnership to facilitate the entry and development of future CGT products in Japan.
Robert Preti, PhD (General Manager of Hitachi Chemical’s Regenerative Medicine Business Sector) and Kazuchika Furuishi, PhD (Deputy General Manager) are joining a leading Japanese management consultant (Colin Novick) to discuss the challenges to consider and examples of solutions for successfully taking your clinical cell or gene therapy program to Japan.
On Wednesday, December 6, the webinar will explore the attractive regulatory changes in Japan, as well as:
- Increased offshore filing activity
- Challenges to consider
- Successful CGT programs brought to Japan
Register now for the Hitachi Chemical Advanced Therapeutics Solutions, LLC sponsored webinar and learn from experts:
http://www.celltherapysociety.org/?page=wbnr_BMI
FDA Lifts Clinical Hold on Cellectis Phase 1 Clinical Trials with UCART123 in AML and BPDCN
Cellectis agreed with the FDA to the following main revisions to be implemented in Phase 1 UCART123 protocols to lift the hold:
- Decrease of the cohort dose level to 6.25×104 UCART123 cells/kg;
- Decrease of the cyclophosphamide dose of the lympho-depleting regimen to 750 mg/m²/day over three days with a maximum daily dose of 1.33 grams of cyclophosphamide;
- Inclusion of specific criteria at Day 0, the day of UCART123 infusion, such as no new uncontrolled infection after receipt of lymphodepletion, afebrile, off all but replacement dose of corticosteroids, no organ dysfunction since eligibility screening;
- Provision to ensure that the next three patients to be treated in each protocol will be under the age of 65;
- Provision to ensure that the enrollment will be staggered across the UCART123 protocols AML123 and ABC123: at least 28 days should elapse between the enrollments of two patients across the two studies.
Mustang Bio Establishes CAR T Cell Therapy Manufacturing Facility in Massachusetts
Facility located in the UMass Medicine Science Park will support clinical development and commercialization of CAR T pipeline
Preparations underway for clinical production in 2018
NEW YORK, Oct. 30, 2017 — Mustang Bio, Inc. (“Mustang”) (NASDAQ:MBIO), a Fortress Biotech, Inc. (NASDAQ:FBIO) company focused on the development of novel immunotherapies based on proprietary chimeric antigen receptor engineered T cell (CAR T) technology, announced today that it has entered into a lease agreement with the UMass Medicine Science Park in Worcester, Massachusetts, for a manufacturing facility to support the clinical development and commercialization of the Company’s CAR T product candidates.
 The facility is expected to be operational for the production of personalized CAR T therapies in 2018. Mustang anticipates initially building cell-processing capabilities to support its lead CAR T product candidates MB-101 in glioblastoma, and MB-102 in acute myeloid leukemia and blastic plasmacytoid dendritic cell neoplasm. [Read more…]
The facility is expected to be operational for the production of personalized CAR T therapies in 2018. Mustang anticipates initially building cell-processing capabilities to support its lead CAR T product candidates MB-101 in glioblastoma, and MB-102 in acute myeloid leukemia and blastic plasmacytoid dendritic cell neoplasm. [Read more…]
- « Previous Page
- 1
- …
- 106
- 107
- 108
- 109
- 110
- …
- 123
- Next Page »