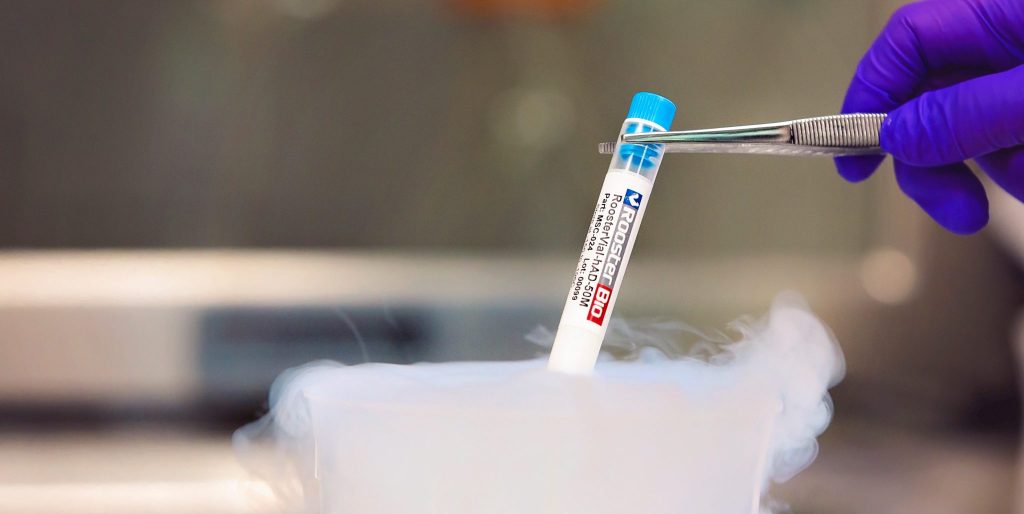
RoosterBio has announced the launch of the Regenerative Medicine (RM) industry’s first xeno-free (XF) hMSC cell banks to advance and simplify stem cell biomanufacturing. By incorporating XF starting materials at early stages, RM scientists can perform more clinically relevant work, resulting in an expedited path towards clinical testing programs. Due of the importance of this announcement, I followed up with Dr. Priya Baraniak, Director of BD & Strategic Marketing at RoosterBio, to further explore details of the launch. [Read more…]
RoosterBio has announced the launch of the Regenerative Medicine (RM) industry’s first xeno-free (XF) hMSC cell banks to advance and simplify stem cell biomanufacturing. By incorporating XF starting materials at early stages, RM scientists can perform more clinically relevant work, resulting in an expedited path towards clinical testing programs. Due of the importance of this announcement, I followed up with Dr. Priya Baraniak, Director of BD & Strategic Marketing at RoosterBio, to further explore details of the launch. [Read more…]
Exopharm to commercialize a new type of nano-drug for regenerative medicine
May 5, 2017, Melbourne, Australia — A private Australian biotechnology company called Exopharm has announced its investment to bring a completely new type of drug called exosomes into local clinical trials.
Exopharm is founded and backed by Dr Ian Dixon, the Melbourne-based technology innovator behind an ASX-listed cell therapy company, the anisina anticancer drug and the Altnia Group incubator. After more than 3 years of secret R&D, Altnia is spinning Exopharm out as a specialist company to translate the promising scientific research around exosomes into routine clinical use worldwide for regenerative medicine. Exopharm will build on its patented purification technology called LEAP to make its proprietary exomeres in large scale and at low cost. [Read more…]
RoosterBio Launches Xeno-Free hMSC Cell Banks to Advance Stem Cell Biomanufacturing
RoosterBio Launches the Regenerative Medicine Industry’s First Xeno-Free hMSC Cell Banks to Advance and Simplify Stem Cell Biomanufacturing
 FREDERICK, MARYLAND, May 04, 2017–At a CAGR of 23.2% and estimated revenue of $68B by 2020, the Regenerative Medicine (“RM”) industry continues to call for new tools designed to accelerate this aggressive growth. Based on strong RM market demand, RoosterBio Inc today announces the launch of xeno-free (“XF”) human bone-marrow derived mesenchymal stem/stromal cell (hMSC) products.
FREDERICK, MARYLAND, May 04, 2017–At a CAGR of 23.2% and estimated revenue of $68B by 2020, the Regenerative Medicine (“RM”) industry continues to call for new tools designed to accelerate this aggressive growth. Based on strong RM market demand, RoosterBio Inc today announces the launch of xeno-free (“XF”) human bone-marrow derived mesenchymal stem/stromal cell (hMSC) products.
Future of Regenerative Medicine with Kyle Cetrulo of AuxoCell
I had the honor of interviewing Kyle Cetrulo, CEO of AuxoCell Laboratories. In this interview, we explore the upcoming Future of Regenerative Medicine Congress, as well as future directions for regenerative medicine at large. Enjoy. [Read more…]
Could Google Be the Next Big Force in Regenerative Medicine?
Google has been massively (and quietly) increasing its presence in the regenerative medicine sector for several years.
 In February 2016, Google’s life sciences business, Verily Life Sciences, announced it was negotiating terms to lease a giant 400,000-square-foot facility formerly occupied by Onyx Pharmaceuticals, a company acquired by Amgen for $9.7 billon in 2013.
In February 2016, Google’s life sciences business, Verily Life Sciences, announced it was negotiating terms to lease a giant 400,000-square-foot facility formerly occupied by Onyx Pharmaceuticals, a company acquired by Amgen for $9.7 billon in 2013.
The facility in Mountain View, California, is about 30 miles north of Alphabet Inc., the holding company for Google (NASDAQ: GOOG). It could support as many as 1,000 Verily Life Sciences employees. Land is also included in lease, a rarity in the Bay Area, allowing Verily the potential for future expansion. [Read more…]
- « Previous Page
- 1
- 2
- 3
- 4
- 5
- …
- 13
- Next Page »