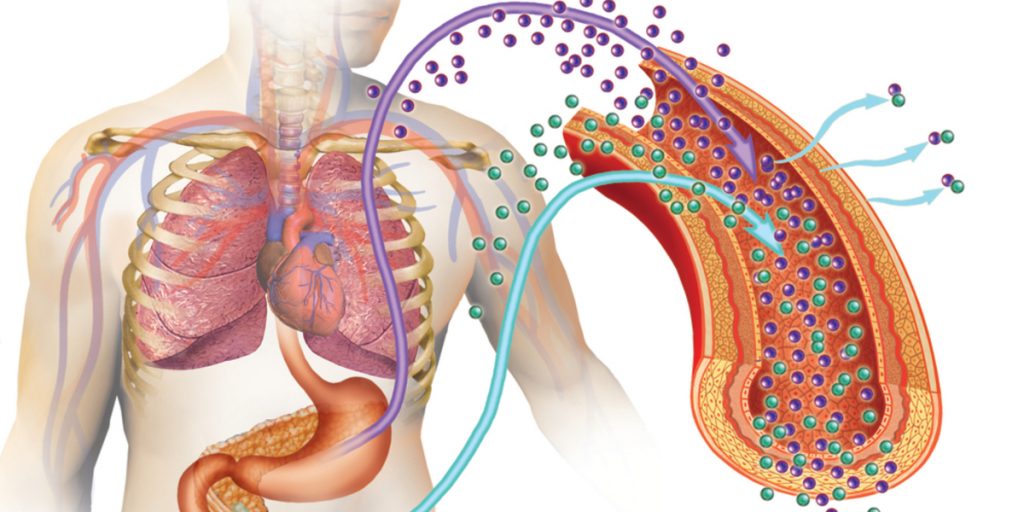
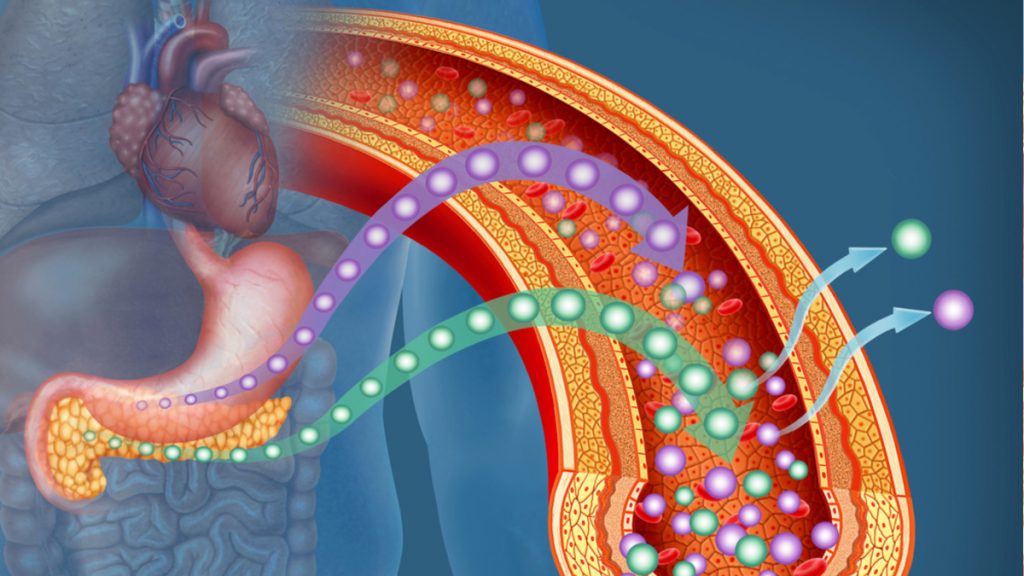
A historic milestone was set on June 28, 2023, when the U.S. Food and Drug Administration (FDA) granted approval for Lantidra to CellTrans, Inc., a company headquartered in Chicago, Illinois. Lantidra is a groundbreaking allogeneic pancreatic islet cellular therapy derived from the pancreatic cells of deceased donors. This landmark decision made it the world’s first cellular therapeutic to be approved for the treatment of type 1 diabetes, a disease that does not yet have a permanent treatment option. Lantridra is designed specifically for adults who have difficulty achieving target glycated hemoglobin levels due to recurring episodes of severe hypoglycemia. [Read more…]
Top Companies Developing Cell Therapy Treatments For Diabetes
Below is a list of the top companies exploring cell therapy treatments for diabetes.
In this article:
- Companies Exploring Stem Cell Treatment for Diabetes
- Seraxis | Cell Replacement Therapy for Type 1 Diabetes
- Unicyte AG | Diabetes Cell Therapy
- ViaCyte | Insulin for Diabetes
- Sernova | Stem Cell for Diabetes Treatment
- Betalin Therapeutics | Insulin Production
- AltuCell | Stem Cell Therapy for Diabetes Type 2
- NextCell Pharma AB | Stem Cell Therapy for Diabetes Type 1
- Osiris Therapeutics | Type 1 Diabetes Control
- Mesoblast | Type 2 Diabetes Treatment
- Evotec and Sanofi | Stem Cell for Diabetes
- Orgenesis | Insulin-Producing Cells
- Semma Therapeutics | Type 1 Diabetes Therapy
- Novo Nordisk | Treatment of Type 1 Diabetes
- Beta-O2 | Artificial Pancreas for Diabetes
- Eli Lilly and Sigilon | Stem Cell for Diabetes
- Pharmacyte Biotech | Encapsulated Human Cells
SERAXIS Inc. publishes clinical trial considerations for a type 1 diabetes stem cell-derived therapy
 GERMANTOWN, Md., Nov. 12, 2018 — SERAXIS Inc., a rapidly growing regenerative medicine company today announced the publication of important trial considerations for SR-01, its pluripotent stem cell-derived islet therapy for insulin-dependent diabetes.
GERMANTOWN, Md., Nov. 12, 2018 — SERAXIS Inc., a rapidly growing regenerative medicine company today announced the publication of important trial considerations for SR-01, its pluripotent stem cell-derived islet therapy for insulin-dependent diabetes.
An analysis of islet transplant trials registered on clinicaltrials.gov was conducted with the goal of extracting insights to apply in the study design of SERAXIS lead candidate, SR-01 for the treatment of type 1 diabetes. The results of this analysis were published this week in Stem Cells Translational Medicine (doi:10.1002/sctm.18-0156). SR-01 is a combination therapy of insulin-producing cells encapsulated in the retrievable biocompatible SeraGraftTM device, designed to prevent immune system access to the cells after implantation. [Read more…]
Unicyte and Collaboration Partner Prof. Camussi Achieve Preclinical Milestone in Diabetes Program
Unicyte, a regenerative medicine unit of Fresenius Medical Care, and its collaboration partner Professor Camussi have published results in Stem Cell Reviews and Reports that a potential therapeutic strategy for diabetes is the transplantation of induced-insulin secreting cells.
The Global Disease Burden of Diabetes Mellitus
Diabetes mellitus is one of the most common diseases globally. Diabetes mellitus (DM), commonly referred to as diabetes, is a group of metabolic disorders in which there are high blood sugar levels over a prolonged period. With an aging population and surging rates of obesity, the incidence of this debilitating disease on the rise. [Read more…]
Technology Licensed by Axolotl Biologix Awarded NIH Grant for R&D of Bioengineered Wound-healing Solution for Diabetes Patients
Research to focus on the development of membrane dressing to accelerate wound closure and improve scar tissue strength and flexibility
(PHOENIX) – Phoenix-based biotechnology company Axolotl Biologix, an innovator in regenerative medicine technologies, announced that technology it has exclusively licensed has been awarded a $224,500 grant funded by the National Institutes of Health (NIH) for the research and development of a wound-healing device for chronic wounds in patients suffering from diabetic foot ulcers. [Read more…]