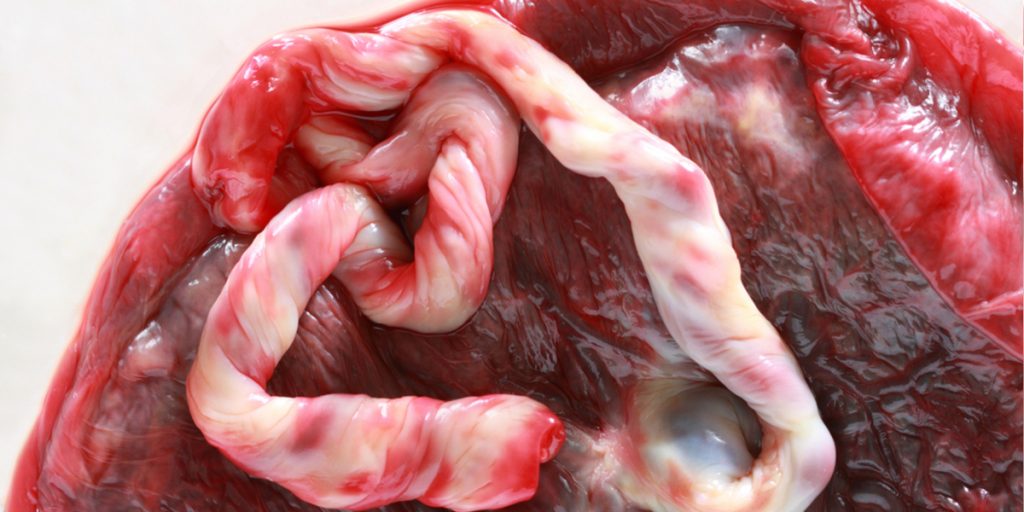
What are placental stem cells and what are placental stem cells used for? As placental stem cells get explored for a variety of therapeutic applications, these questions become increasingly important. The opportunity to collect placental stem cells is also a once in a lifetime opportunity. [Read more…]
Celularity – Big Bucks, Billionaire Investors, Bold Dreams
Celularity is a biotechnology company that specializes in leveraging biologically active cell populations within the postpartum human placenta. Founded on the pioneering work of Robert Hariri, MD, PhD, in human placenta-derived cellular therapeutics and biomaterials, Celularity is uniquely positioned to harness the potential of most biocreative event on earth: human birth. [Read more…]
Dr. Robert Hariri – Placental Pioneer Launches Celularity with $250M Cash

Dr. Robert Hariri is a fascinating figure in the stem cell industry, because he was the earliest pioneer to recognize the regenerative potential of the human placenta. In August 2017, Hariri brought his latest company, Celularity, into the spotlight by announcing a successful Series A financing with contributions from influential investors, including United Therapeutics Corporation, Celgene, Sorrento Therapeutics, and Human Longevity, Inc. (HLI).
Today, Celularity’s investor contributions have reached an astounding $250 million, including contributions from the Genting Group, Dreyfus Family Office and Tony Robbins, among other notable investors.
Celularity, Inc.
Celularity, a spin-out company from global biopharmaceutical giant Celgene, announced this morning that it has raised $250 million to support development of revolutionary placental-derived products, including T-cells that will be immune advantaged because of their derivation from the placenta. This is a massive accomplishment, because it dwarfs the $225 million in Series A financing raised by BlueRock Therapeutics in December 2016 that was previously a landmark accomplishment within the stem cell industry.
With the Celgene snagging Juno Therapeutics for a shocking $9 billion in January 2018 and Gilead picking up Kite Pharma for an astounding $11.9 billion in August 2017, Celularity is also joining the ranks of powerful immunotherapy companies that could be a target for merger or acquisition.
If that wasn’t enough, Celularity is looking to burst a major bottleneck in the industry by deriving T-cells from a single (allogeneic) cell line, potentially positioning itself to slash the price point for CAR-T treatments. Currently, Gilead and subsidiary Kite Pharma have a CAR-T cell therapy on the market (Yescarta) with a price point of $373,000 per patient and Novartis has a genetically modified autologous CAR-T therapy (Kymriah) priced at $475,000 per treatment.
At its core, Celularity is a biotechnology company that specializes in leveraging biologically active cell populations within the postpartum human placenta. Founded on the pioneering work of Robert Hariri, MD, PhD, in human placenta-derived cellular therapeutics and biomaterials, Celularity is uniquely positioned to harness the potential of most biocreative event on earth: human birth. [Read more…]
Celularity and GX Acquisition Corp. Announce Merger Agreement to Create a Publicly Listed Leader in Allogeneic Cellular Therapy
FLORHAM PARK, N.J. and NEW YORK, Jan. 8, 2021 — Celularity, Inc. (“Celularity”), a clinical-stage biotechnology company, leading the next evolution in cellular medicine with the development of off-the-shelf allogeneic therapies derived from the postpartum human placenta, and GX Acquisition Corp. (Nasdaq: GXGX), a special purpose acquisition company, today announced they have entered into a definitive merger agreement pursuant to which GX Acquisition Corp. will combine with Celularity. Upon the closing of the transaction, which is expected to occur in the second quarter of 2021, GX Acquisition Corp. will be renamed Celularity Inc., and its common stock and warrants are expected to remain listed on Nasdaq under the new ticker symbols “CELU” and “CELUW.” [Read more…]
How Celularity is Leading the Next Evolution in Cellular Medicine
Celularity is a clinical-stage cell therapeutics company delivering transformative allogeneic cellular therapies derived from the postpartum human placenta. Using proprietary technology in combination with its IMPACT™ platform, Celularity is the only company harnessing the purity and versatility of placental-derived cells to develop and manufacture innovative and highly scalable off-the-shelf treatments for patients with cancer, inflammatory and age-related diseases.
Founded on the pioneering work of Robert Hariri, MD, PhD, in human placenta-derived cellular therapeutics and biomaterials, Celularity is uniquely positioned to harness the potential of most biocreative event on earth: human birth. [Read more…]