|
|
What do we know of the safety and efficacy of stem cell therapeutics? Over 50 years have passed since the discovery of hematopoietic stem cells (bone marrow transplantation to cure diseases such as leukemia), over 20 years since the discovery of human embryonic stem cells (hESC), and 15 years since the discovery of induced pluripotent stem cells (iPS cells). There are now thousands of stem cell trials underway in clinics worldwide.
Despite this experience, what do we actually know about the safety and efficacy of stem cell therapeutics?
Stem Cell Therapeutics and Safe Practice
In this article:
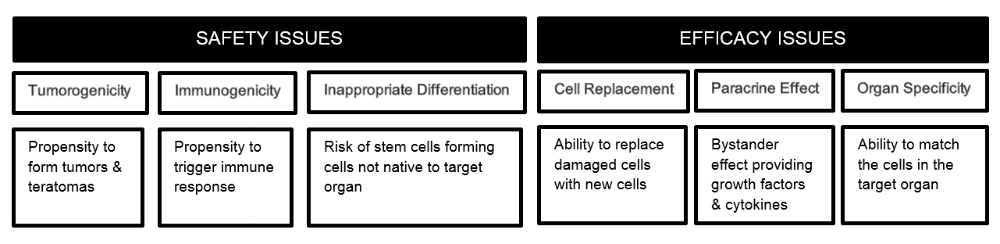
Safety and Efficacy Issues with Stem Cell Therapies
In this novel and increasingly complex stem cell space, it is perhaps a timely question to ask, especially since the regulatory assessment criteria designed for conventional medicines are not directly applicable for stem cell therapy.
Tumorigenicity in Cellular Therapeutics
Tumorigenicity is a major concern with stem cell therapeutics. Delivery of a cell with unlimited potential for renewal and capacity to differentiate into any human cell type carries a safety concern not associated with any other class of therapy. This is a significant and well-accepted issue for hESC and iPS cells as there is growing evidence that their derivatives have similar tumorigenic risks. Even cells derived from autologous iPS cells once considered safe have recently begun to cause concern. A clinical trial using autologous iPS cell-derived RPEs (retinal pigment epithelium) was halted in 2015 after only one person had received a treatment. As the lab prepared to treat the second trial participant, two small genetic changes were detected in both the patient’s iPS cells and the RPE cells derived from them.
Growing Problems of an Immune Response
Immunogenicity is a growing concern and one that remains to be fully elucidated as available scientific evidence is still notoriously contradictory. Although there are reports of immuno-privileged status for some therapeutic stem cells, such as mesenchymal stem cells (MSC) (Drukker and Benvenisty, 2004), any foreign cell introduced into a patient is subject to immune surveillance (Swijnenburg et al., 2008) and will trigger an immune response. Immunogenicity of allogeneic cells is well known, but there is growing evidence that even autologous and syngeneic cell transplants from hESC and iPS cells and their derivatives carry a significant degree of immunogenicity (Gao et al., 2016).
Immunosuppression: attempting to cope with immunogenicity results in significant health risks
Many advocate the use of immunosuppression to get around the immune-rejection of all allogeneic stem cell transplants, but this is starting to become medically questionable. Long-term immunosuppression is not an option due to its inherent adverse side-effects that can even lead to fatality; in addition, patients already weakened by the underlying disease/injury or aging could end up with significant health issues (infections) with a long-term administration of immunosuppressants.
Negative Reaction to Cell Therapy Treatment
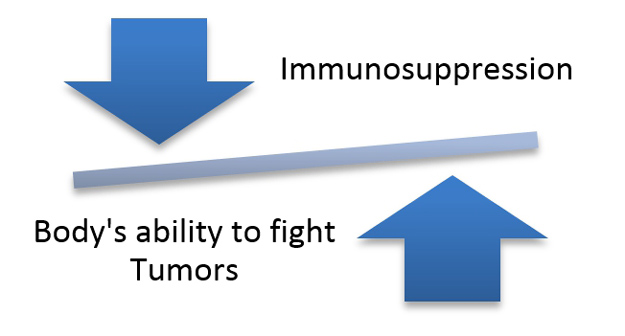
For pluripotent cell-derived implants, to suppress the immune system of the recipient at the same time as implanting hESC or iPS cells or their functionally relevant derivatives with a known propensity for tumorigenesis exposes patients to an unacceptable level of risk, since a suppressed immune system is not able to effectively fight off tumors. Finally, safety issues linked to immunogenicity may only allow the patient to be injected safely once in their lifetime with allogeneic cells, as there is evidence that repeated dosing with allogeneic cells could result in a strong immune response against the cells and even anaphylaxis.
Allogeneic Stem Cell Issues
Immunogenicity and tumorigenicity are tightly linked through an “unfavorable” balance. This is a fundamental safety issue for allogeneic stem cells. On the other hand, with autologous stem cell treatment, the patient’s immune system remains intact and retains its ability to fight off tumors, making autologous stem cell treatment safer.
Inappropriate Differentiation
Inappropriate Differentiation is a concern with stem cells derived from a different lineage than the target tissue or organ where these cells are transplanted. For example, MSC (which differentiate into bone, cartilage, muscle and fat cells) implanted into another tissue or organ such as the heart or brain carry a risk of growing into bone, cartilage and/or fat tissue. In one published case, a woman that was injected with MSCs around her eyes ended up with bone tissue growing inside her eyelids.
Efficacy Issues with Implanted Cells
Potency or efficacy comes from Cell Replacement (“seed” effect) and Paracrine (“bystander” or “fertilizer” effect) effects of the implanted stem cells. It is important to note that the bystander effect only goes as far as providing growth factors, and cytokines, and other paracrine effects to the host’s existing cells. In cases where significant tissue has been destroyed or is irreparable, the paracrine function will have only limited or no effect and only stem cells that can graft and replace cells will provide regrowth of tissue and restore functionality.
The Trouble with Paracrine Signals
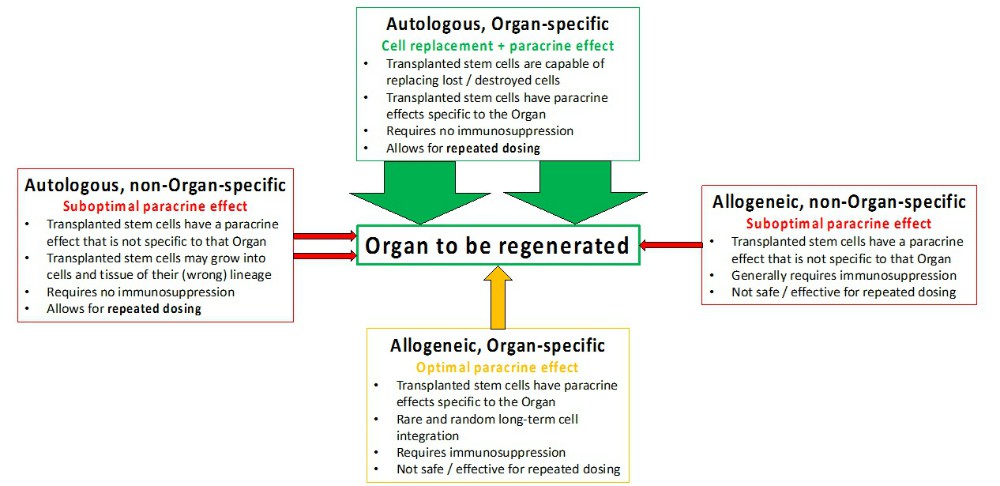
Organ Specificity is increasingly important for safe and effective tissue regeneration, which only autologous organ-specific stem cells can ensure. Even organ-specific allogeneic stem cells pose significant safety issues, which need to be weighed against their efficacy limited solely to the bystander effect.
While autologous stem cells from a different organ may be safe in many circumstances, their efficacy through paracrine signals including growth factor and cytokines secretion will be limited to the non-specific bystander effects. The paracrine effect is specific to the source tissue/organ (such as bone marrow), instead of the organ that they are implanted into. Therefore, the hopes of a universal donor and an off-the-shelf product (allogeneic non-organ specific) would be exceedingly difficult to implement.
Since harvesting organ-specific stem cells in an autologous manner have its limitations, especially for CNS, direct reprogramming of somatic cells is the optimal method for generating autologous organ-specific stem cells.
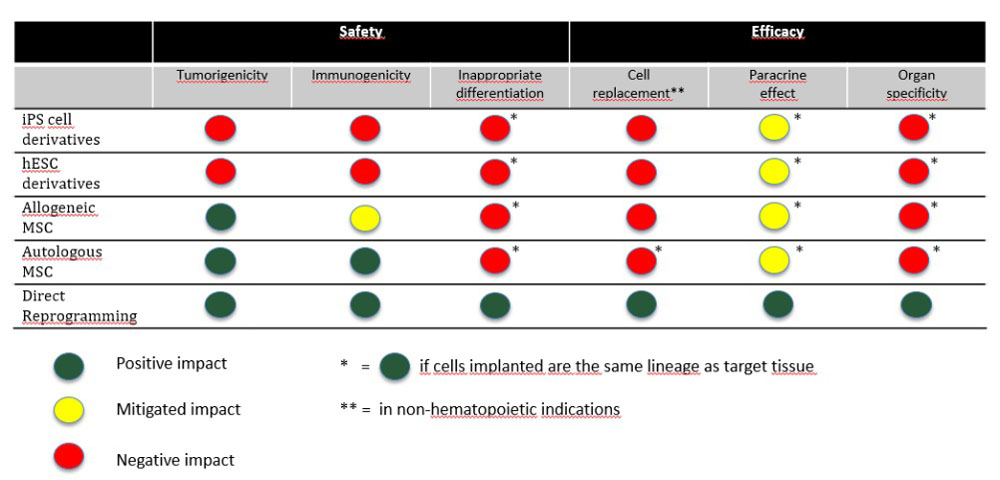
How do Different Stem Cell Therapies Stack up Against these Critical Factors?
Increased understanding of hESCs and iPSCs and their progeny are challenging the possibility of translating them into viable stem cell therapeutics. Similarly, MSCs have extended their therapeutic promises beyond their own natural capabilities, and these goals are proving to be not only ambitious but often unrealistic, similar to expecting a liver transplant to work like a kidney transplant on a failed kidney.
hESC and Their Progeny: Immunogenic and Tumorigenic
hESC and their progeny have been long known to have risks of tumor/teratoma formation, as well as high immunogenicity. In a key study by Robertson et al., a panel of genetically identical mouse embryonic stem cells that differed from recipient mice only at defined genetic loci resulted in a strong immune response and rejection even with just a single MHC class I gene being different, and the speed of rejection was only slightly slower than with fully allogeneic embryonic stem cells.
It would, therefore, seem like it will be nearly impossible to create universal stem cell banks (even with all the combinations of MHC class antigens) that will not be rejected by the recipient patients (Robertson et al., 2007).
iPS cells and Their Progeny: Tumorigenic and Immunogenic, even if Autologous
Surprisingly, and probably less obvious, is the growing evidence that autologous (and syngeneic) iPS cells and their derivatives are immunogenic. “The immunogenicity associated with iPS cells and their progeny seems to be linked to the ectopic expression of certain genes that encode what might best be described as ‘developmental antigens.’
Pluripotency is associated with a pattern of gene expression that is normally lost shortly after implantation, precluding any participation of the associated gene products in the selection of the T-cell repertoire and the induction of immunological tolerance once thymus organogenesis has occurred later in ontogeny” (Fairchild et al., 2016).
For example, Zhao et al. identified nine such genes that fulfilled the criteria of being strongly upregulated upon reprogramming to pluripotency, but failing to be properly silenced upon subsequent differentiation in vitro ( Zhao et al., 2011).
Loss of Potential in iPS Cells
Gao et al. further found that iPS cells and their derivatives lose the memory of their origin and are no longer fully recognized as self by the host’s immune system (Gao et al., 2016). Thus the body no longer recognizes these cells as “autologous”, indicating that it is probably not possible to produce autologous cells via the iPS cell route as was originally hoped.
MSCs: Safer…but What About Efficacy?
In contrast to pluripotent cells (ESCs and iPSCs) and their progeny, harvested MSCs, with limited potential for proliferation and differentiation, are considered “safe” as they can hardly form tumors in vivo (Goldring, et al., 2011). Originally viewed as a potential off-the-shelf product for stem cell therapeutics, there is increasing evidence of the inherent immunogenicity of MSCs and thus immune-rejection by the recipient.
Major histocompatibility complex (MHC)-class I and II molecules are expressed in MSCs, albeit weakly, which makes them potentially less immunogenic than hESC and iPS cells (Gebler, et al., 2012), but they are still immunogenic.
MSCs Replace Tissue in Their Own Lineage
In one published case, Thangakunam et al. found that a repeat injection of the same MSCs resulted in anaphylactic shock in a patient. MSCs are not functionally prepped for all bodily functions, and despite popular beliefs, can only replace tissue of their own lineage: bone, cartilage, and fat.
Such tissue replacement and repair require the MSCs to be autologous (the patient’s own cells), and not allogeneic (a donor’s cells); if the cells are allogeneic then they will only provide trophic paracrine support for limited innate regeneration ( “bystander” effect) until the cells are destroyed by the patient’s immune system.
The Limitation of MSCs in Differentiation
MSCs do not have the ability to replace cells in other organs (e.g. brain, liver or heart) where cells are derived from other lineages, and even the bystander paracrine effect in such organs is limited as it is not specific to those organs. General lack of awareness surrounding this feature has resulted in confusion, whereby, for example, MSCs have been implanted into numerous unrelated tissues (e.g. the brain), with expected disappointing limited beneficial effects.
(As mentioned above, this approach can also be harmful, as e.g. the one case where a patient that received an MSCs injected injection into her eye area that started growing bone inside her eyelids).
Direct Cell Reprogramming for Highest Safety and Efficacy
Autologous multipotent stem cells produced by direct reprogramming may represent the first safe and fully functional therapeutic stem cells (Gao et al., 2016). Gao et al. described “induced pluripotency” and its associated developmental gene expression profiles constitute the major difference in immunogenicity and tumorigenicity between “autologous” iPSCs and directly reprogrammed autologous stem cells.
Low Chances of Tumorigenicity and Immunogenicity
Autologous multipotent stem cells produced by direct reprogramming technology do not have a propensity for tumor formation, nor do they stimulate the immune system. Hence, such autologous reprogrammed cells can be injected several times into a patient, allowing for a treatment regimen that could potentially fully regenerate the target tissue or organ. Being autologous, they will not require immunosuppression.
Directing Cell Differentiation
Furthermore, the differentiation potential of multipotent stem cells is limited to their own specific lineage: a neural stem cell will only have the ability to form neural tissue through differentiation to astrocyte, oligodendrocyte, and neurons. The process of direct reprogramming grants the ability to tailor-make stem cells to the right organ and tissue.
Being made for the right organ, these multipotent stem cells have the capacity to regenerate through Cell Replacement (“seed” effect) and Paracrine or bystander (“fertilizer” effect) effects.
The Importance of Direct Reprogramming
Autologous organ-specific stem cells are the stem cells that were originally intended by many scientists as potentially capable of significant therapeutic effects and even cures. Many placed their hopes on iPS cells and on their potential ability to produce autologous organ-specific stem cells, and 10 years later, iPS cells have found their place as research workhorses, but not into the realm of stem cell therapeutics.
With limited MSC-derived products making it through clinical trials, and the ethical and immunogenic issues linked to fetal and embryonic cells, stem cell scientists are now turning toward direct reprogramming as a way of producing autologous organ-specific therapeutic stem cells.
The Future of Stem Cell Therapeutics
The stem cell industry is at a junction where regulators are attempting to pave the way for safe and efficacious stem cell therapeutics while ensuring patients with speedy access to treatment. With increasing interest and evidence, directly reprogrammed autologous stem cells are expected to make their way to the clinic as the most promising therapeutic stem cell product.
If you found this blog valuable, subscribe to BioInformant’s stem cell industry updates.
What are your thoughts on the safety of stem cell therapeutics being developed by cell therapy companies? Share your thoughts below.



















Nice summary but it avoids a few issues. You seem to imply that successful and non-problematic stem cell therapy will be limited, by and large, to the use of autologous stem cells or reprogrammed cells obtained from these. Are you then implying that cell therapy won’t be useful. In other words, you seem to be saying that the future lies in reprogramming the cells that are already there (which I happen to agree with).
Is this what you are getting at?
I would like to know your thoughts and any data you may have regarding placental tissue, and specifically it’s use in the treatment of soft tissue degeneration or musculoskeletal issues…i.e. rotator cuff, meniscal tears etc. My understanding is that this tissue is immune privileged and carries with it no chance of rejection along with being non-tumorigenic. Placental tissue is touted as having within it an extracellular matrix comprised of the necessary biomolecules, growth factors along with a collagen scaffold capable of tissue remodeling/knitting.
Thank you
The entire team is amazing. Stem Cell of America has given my chronic pain of peripheral neuropathy a run for its money. It’s been a process but I am now able to walk without limping, sit without hurting and actually play with my dog for more than 5 mins.
when will there be stem cell treatment for generalized idiopathic epilepsy ?
Hi Cade, thanks for your articles and all you do at BioInformant. I think you’re awesome and a positive force for advancement of regenmed.
And yet forgive me for wondering whether framing the questions of cell types like this is akin to asking how a human can fare against a dog in a scent detection contest. Or how well a pony can fare against humans to win the role of MacBeth at a Shakespeare festival… Or asking me to hit the rugby field against 10 year veterans of the sport … etc.
In other (here unspecified) categories, each of these cell types in the chart can greatly excel. The industrial scalability of allo-MSCs is amazing, for example, though still not fully realized. And they can be manipulated in so many ways to carry (or secrete via EVs) genetic medicines–also a possibility barely realized.
Just getting this off my chest, you understand. Keep fighting the good fight.