Boston BioLife (www.bostonbiolife.com) is a cutting-edge organization that provides workshops for physicians and scientists interested in learning regenerative medicine. Its mission is to facilitate the introduction of emerging life sciences technologies to scientists and healthcare providers. [Read more…]
Boston BioLife (www.bostonbiolife.com) is a cutting-edge organization that provides workshops for physicians and scientists interested in learning regenerative medicine. Its mission is to facilitate the introduction of emerging life sciences technologies to scientists and healthcare providers. [Read more…]
Pall Corporation and RoosterBio to Co-Market Mesenchymal Stem/Stromal Cells and Bioreactors
The agreement will encompass a co-development and marketing partnership.

OCTOBER 05, 2016 –Pall Corporation, a global leader in bioprocess equipment and disposables, announced the signing of a commercial collaboration agreement with RoosterBio Inc, an innovative adult stem cell technology company. The agreement will encompass a co-development and marketing partnership, delivering complete solutions for industrial-scale bioreactor expansion of human Mesenchymal Stem/Stromal Cells (hMSCs). [Read more…]
History of Stem Cells | Mesenchymal Stem Cells
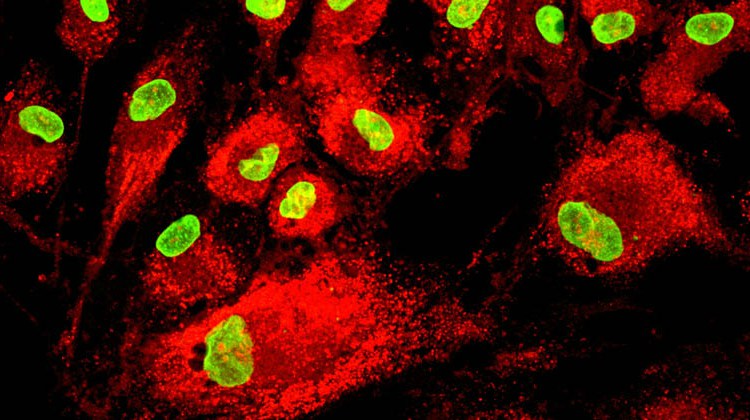
Mesenchymal stem cells (MSCs) are multipotent stem cells that can differentiate into a variety of cell types, including osteoblasts, chondrocytes, myocytes, and adipocytes. MSCs are of intense therapeutic interest, because they represent a population of cells with the potential to treat a range of acute and degenerative diseases. MSCs are advantageous over other stem cells types for a variety of reasons, including that they are immunoprivileged, making them an advantageous cell type for allogeneic transplantation.
Allogeneic means that cells from a donor are used in the treatment of a different person.
Overview of Mesenchymal Stem Cells (MSCs)
In addition to secreting factors that can stimulate tissue repair, MSCs can substantially alter their microenvironment, exerting effects that are both anti-inflammatory and anti-fibrotic. MSCs are also advantageous over other stem cells types, because they avoid the ethical issues that surround embryonic stem cell research and can positively impact immune function. Because of their unique capacity to form structural tissues, MSCs also being explored for use in 3D printing applications.
History of Mesenchymal Stem Cells (MSCs)
1900s
The discovery of mesenchymal stem cells (MSCs) dates back to the early 1900’s.
1924
In 1924, the Russian-born researcher Alexander Maximow used histology to identify a type of precursor cell within the mesenchyme that could differentiate into a variety of blood cell types.[1] While the term mesenchymal stem cell did not exist, this is the earliest known reference to the cell type.
1960s
Nearly 40 years later, in the 1960’s, researchers Ernest McCulloch and James Till identified the clonal qualities of marrow cells. [2],[3] It then took another decade until an ex vivo assay was developed that allowed for examination of the clonal nature of multipotent marrow cells.[4] [Read more…]
Physical And Functional Characteristics Of Mesenchymal Stem Cells (MSCs)
Mesenchymal stem cells (MSCs) are multipotent cells that can differentiate into a variety of cell types, including bone, cartilage, muscle and fat cells, among other cell types. MSCs are a leading type of adult stem cell being investigated for therapeutic applications, because they have the potential to treat a range of acute and degenerative diseases.
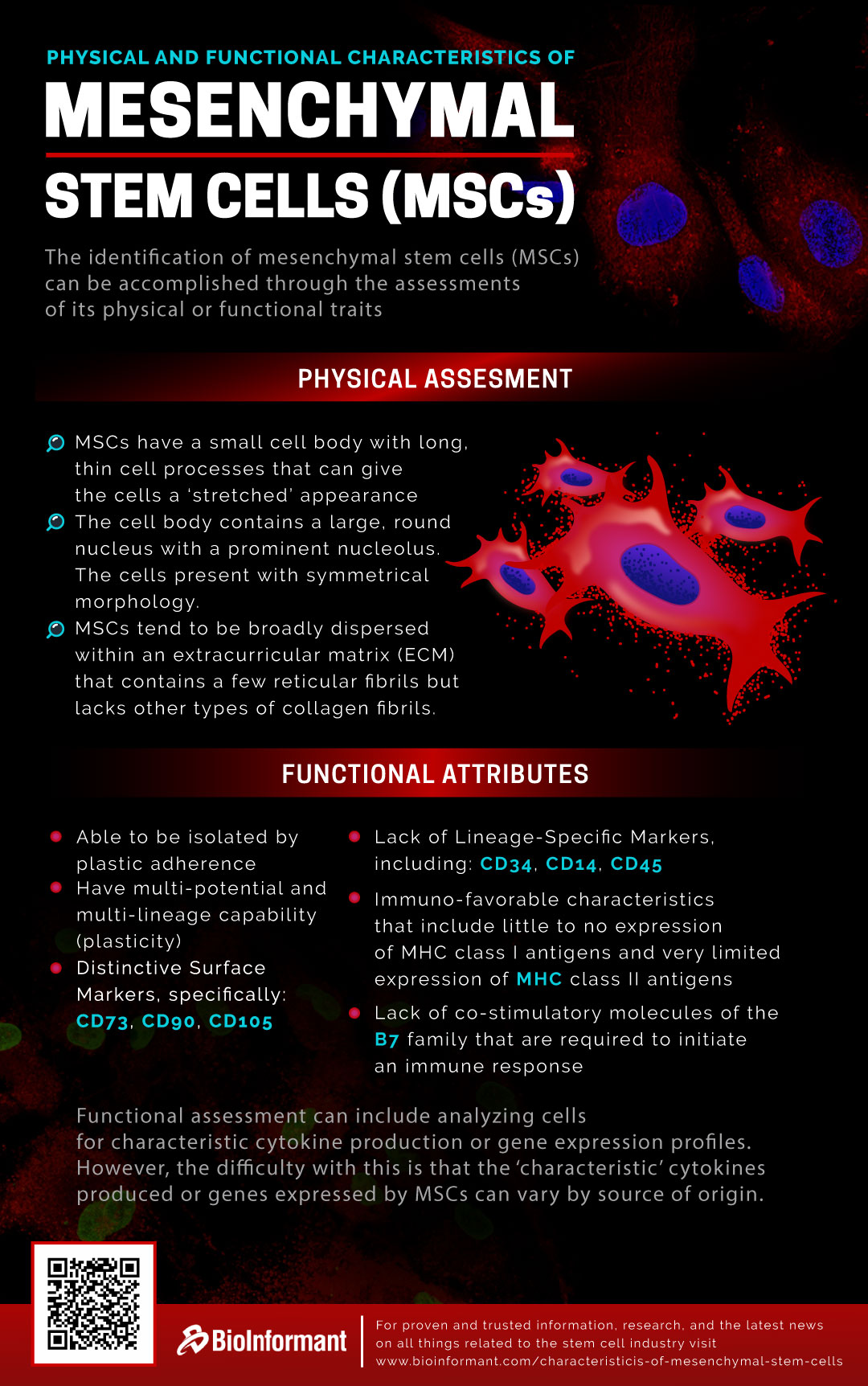
Characteristics of Stem Cells | Identification of MSCs
The identification of mesenchymal stem cells (MSCs) can be accomplished through the assessment of either physical or functional traits.
Physical Assessment of Mesenchymal Stem Cells (MSCs)
The first and most convenient approach to identifying MSCs is through physical assessment. Morphologically, MSCs have a small cell body with long, thin cell processes that can give the cells a “stretched” appearance.
The cell body contains a large, round nucleus with a prominent nucleolus. The cells present a symmetrical morphology.
MSCs also tend to be broadly dispersed within an extracellular matrix (ECM) that contains a few reticular fibrils but lacks other types of collagen fibrils.
Functional Attributes of Mesenchymal Stem Cells (MSCs)
Another secondary approach for identifying mesenchymal stem cells is through functional assessment.
Criteria for functionally identifying MSCs are as follows:
- able to be isolated by plastic adherence
- have the multi-potential and multi-lineage capability (plasticity)
- distinctive surface markers, specifically: CD73, CD90, and CD105[1]
- lack of lineage-specific markers, including CD34, CD14, and CD45[2]
- immuno-favorable characteristics that include little to no expression of MHC class I antigens and very limited expression of MHC class II antigens[3]
- lack of co-stimulatory molecules of the B7 family that are required to initiate an immune response
In addition, functional assessment can include analyzing cells for characteristic cytokine production or gene expression profiles. However, the difficulty with this approach is that the “characteristic” cytokines produced or genes expressed by MSCs can vary by source of origin. For instance, bone marrow-derived MSCs and adipose-derived MSCs exhibit different, source-specific, cytokine production.[4]
Save this infographic for later!
Footnotes
[1] Klingemann H, et al. Mesenchymal Stem Cells, Sources and Applications. Transfusion Medicine and Hemotherapy 2008; 35: 272-277.
[2] Friedman R, Betancur M, Tuncer H, Boissel L, Klingemann H. Umbilical cord mesenchymal stem cells: Adjuvants for human cell transplantation. Biol Blood Marrow Transplant 2007; 13: 1477–1486.
[3] Friedman R, Betancur M, Tuncer H, Boissel L, Klingemann H. Umbilical cord mesenchymal stem cells: Adjuvants for human cell transplantation. Biol Blood Marrow Transplant 2007; 13: 1477–1486.
[4] Gonzalex-Ray, et al. Human adipose-derived mesenchymal stem cells reduce inflammatory and T cell responses and induce regulatory T cells in vitro in rheumatoid arthritis. Ann Rheum Dis 2010; 69: 241-248.
What are your thoughts about the characteristics of stem cells, mesenchymal stem cells to be specific? Let us know in the comments section below.
Up Next: What Diseases Are Treatable With Hematopoietic Stem Cells (HSC) From Cord Blood?
CDI Launches Opsis, Provia Expands Perinatal Storage, CCBC Gets Biologics License
We have released coverage of this week’s stem cell industry activity that I am excited to share with you. It was a major week for stem cells, with CDI launching Opsis Therapeutics, Provia Laboratories expanding its stem cell product portfolio, CCBC getting a FDA Biologics License for CLEVECORD™, STEMCELL Technologies exploring genomic instability in an upcoming webinar, and iPSCs turning 10. [Read more…]
- « Previous Page
- 1
- …
- 122
- 123
- 124
- 125
- 126
- …
- 194
- Next Page »