Stem Cell News
BioInformant's coverage of stem cell news includes iPS cells, mesenchymal stem cells, hematopoietic stem cells, neural stem cells and more.
As the first and only market research firm to specialize in stem cell news, BioInformant research has been cited by the Wall Street Journal, Xconomy, AABB, and Vogue Magazine.
Serving Fortune 500 leaders that include GE Healthcare, Pfizer, and Goldman Sachs, BioInformant is your global leader in stem cell industry news.
GMP cell CDMO I Peace, Inc. obtains accreditation as ISO 17025: 2017 compliant
Starts undertaking quality evaluation and characterization of iPSCs in accordance with ISO standards
PALO ALTO, Calif., Dec. 6, 2023 — Leading GMP cell CDMO I Peace, Inc., specializing in induced pluripotent stem cells (iPSCs) and iPSC-derived cell therapies, announced that its Palo Alto quality control testing facility has been accredited as ISO 17025: 2017 compliant. The accreditation was certified by ANSI National Accreditation Board, one of the leading accreditation bodies in the world. The tests in the scope of accreditation center on the characterization of iPSCs and include cell count and viability per USP <1046>, detection of pluripotency marker expression by flow cytometry per USP <1027>, and also by immunofluorescence assay. [Read more…]
Seattle Children’s launches BrainChild Bio to accelerate CAR T-cell therapies for children with brain tumors
BrainChild Bio’s initial CAR T-cell therapy program will focus on advancing ongoing clinical trials evaluating safety and efficacy in pediatric brain tumors.
December 5, 2023, Seattle, WA – Seattle Children’s announced the launch of BrainChild Bio, Inc. to accelerate the advancement of chimeric antigen receptor (CAR) T-cell therapies in Central Nervous System (CNS) tumors. BrainChild Bio will be granted an exclusive license to novel CAR T-cell technology for CNS tumors developed at Seattle Children’s and will build upon the pioneering CAR T-cell therapy and clinical translational work of Michael Jensen, M.D., and his team at Seattle Children’s Therapeutics. Seattle Children’s has provided the initial equity funding for BrainChild Bio which will operate as an independently managed corporation. [Read more…]
StemSight Bolsters Leadership Team and Adds Extensive Experience in Cell Therapy Commercialization with Key Hire
In the field of groundbreaking regenerative medicine, StemSight’s aim to cure blindness takes a major leap forward with the strategic appointment of Dr Ross Macdonald as Chief Commercial Officer.
Tampere, Finland, 21.11.23 — StemSight Oy, the leading deep tech company dedicated to eliminating corneal blindness through stem cell technology, proudly announces the appointment of Dr Ross Macdonald to advance the Company’s commercial strategy. With over three decades of pharmaceutical and biopharma industry expertise, Macdonald’s inclusion in StemSight’s team brings a new breadth of experience to the Company. [Read more…]
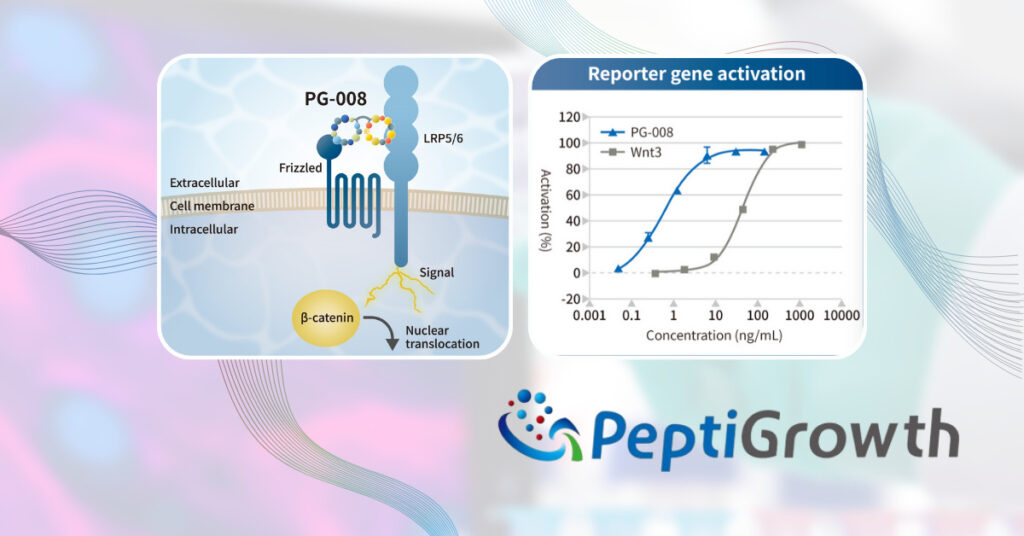
PeptiGrowth Inc. is Launching a Novel Synthetic Peptide Wnt3a alternative peptide (β-catenin pathway agonist)
PeptiGrowth Inc. (Headquarters: Chiyoda-ku, Tokyo, President: Jiro Sugimoto) has successfully developed a novel synthetic peptide called “Wnt3a alternative peptide (β-catenin pathway agonist)” which has equivalent function to recombinant Wnt3a as well as GSK-3β inhibitors, such as CHIR99021(CHIR)). This product will be available in the middle of November 2023. [Read more…]
- « Previous Page
- 1
- …
- 5
- 6
- 7
- 8
- 9
- …
- 122
- Next Page »