|
|
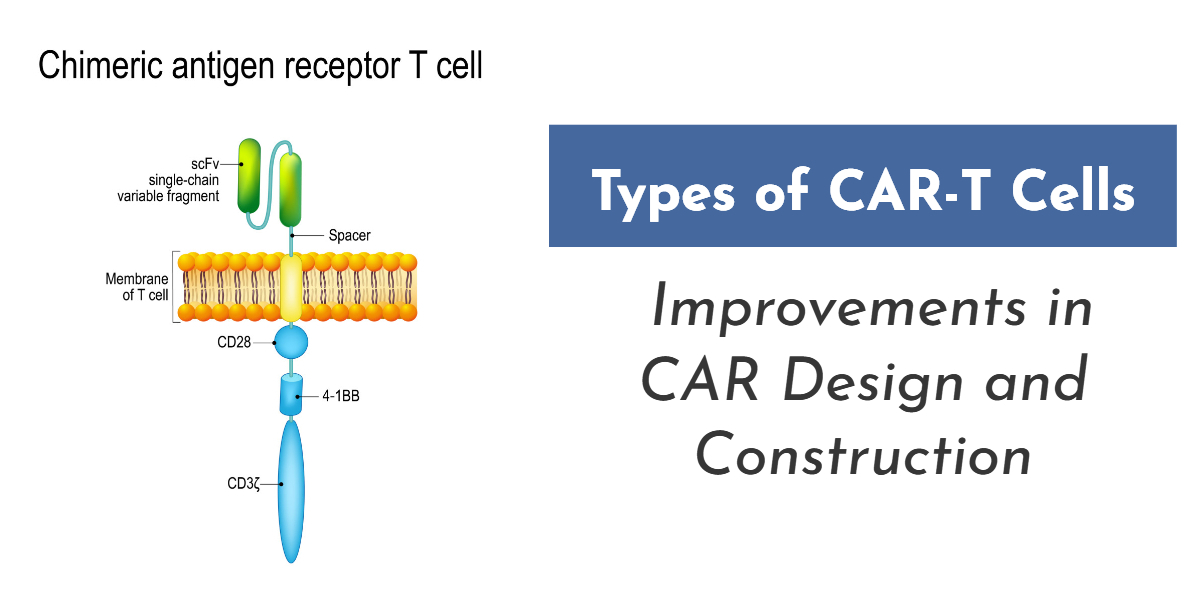
What are the types of CAR-T cells and how have they changed in recent years? CAR-T cells are immune cells that are artificially engineered to express a particular chimeric antigen receptor (CAR), which have shown great potential in killing cancer cells. Chimeric antigen receptors (CARs) are artificially produced recombinant receptors having both antigen-binding and T-cell activating potentials.
CARs are composed of an extracellular recognition domain sourced from monoclonal antibodies (mAbs) and an intracellular signaling domain including CD3- and costimulatory domains for triggering endogenous signaling pathways of T-cells.
The recognition domain of the CAR can target specific antigen molecules expressed on the surface of cancer cells and thereby triggering unique responses including activation and killing activity of T-cells mediated by the signaling domain.
First Generation CAR-T Cells
In the first generation of CAR-T, the intracellular domain consists of CD3 ζ- chain or FcεRIγ, and it is the primary transmitter of signals from endogenous T cell receptor (TCR). But, these CAR-T cells were not able to produce enough interleukin-2 (IL-2), so in order to kill tumor cells, researchers had to administer exogenous IL-2.
Thus, the first generation of CAR-T cell therapy transfected with single-chain receptors were effective only when exogenous cytokine was administered into the patients. Most of the initially conducted studies with first-generation CAR-T cells could not achieve the desired effects because of inadequate proliferation, a short life span in vivo and inadequate secreted cytokines.
Second Generation CAR-T Cells
In the construction of second generation CAR-T cells, additional signaling domains were added. The intracellular domain consisted of T-cell receptors (TCR), cytokine receptors and costimulatory receptors. The first signal activated by the TCR recognized the antigenic peptide-MHC complex on the surface of tumor cells. The signal from the co-stimulatory signal, produced by a costimulatory molecule such as CD28/B7 promoted the IL-2 synthesis to complete the activation of T cells and avoid apoptosis.
Thus, it was found that CARs with only the CD3ζ in the intercellular domain cannot activate CAR-T cells without a costimulatory signal. Therefore, second generation CARs were constructed by adding intracellular signaling domains from various costimulatory protein receptors such as CD28 or CD137 (4-1BB and CD134 (OX40)) in order to provide additional signals to the T cell, which improved the proliferation, cytotoxicity, and sustained response, and prolong the life of CAR-T cells in vivo.
CD28-mediated co-stimulation is very much essential for the regulation of proliferation and survival for lymphocytes, and plays a vital role in the establishment of memory cells and effector cells. CD134 is essential for sustaining proliferation and strengthening IL-2 production. CD137 is able to maintain the response signal of T cells, which plays a vital role in the survival of T cells and the memory of CD8+ T-cells.
Third Generation CAR-T Cells
Typically, the third generation CARs is composed of an antigen binding unit, a spacer (hinge), a transmembrane domain and endodomain.
The major difference between the third generation and other generations is the co-stimulatory signaling domain, which is made up of two different co-stimulatory signaling units in the intracellular regions along with the CD3ζ or FcεRIγ signaling domain.
The greatest advantage of the third generation CARs is that it can effectuate high level cytokine secretion and considerably increase proliferation speed and survival rate of engrafted T cells. Scientists have made a range of CARs with signaling components such as CD28, CD137 (4-1BB), CD134 (OX40) and DAP10.
Fourth Generation CAR-T Cells
The fourth-generation CARs were constructed by adding IL-12 to the base of the second-generation constructs, and were also named as T cell redirected for universal cytokine-mediated killing (TRUCKs).
TRUCKs strengthen T-cell activation and activate and attract innate immune cells to destroy antigen-negative cancer cells in the targeted tumor.
The fourth generation CAR-T cells are constructed in such a way that they store transgenic cytokine in CAR-T cell itself and only released when it is induced. Moreover, they are also constructed by incorporating caspase-9 gene as a suicide gene which can be activated to rapidly withdraw CAR-T cells once the anti-tumor effect is achieved.
To summarize, the design of CAR-T cells has continually evolved, with new and novel approaches being incorporated over time. What changes do you anticipated will come next? Share your thoughts in comments below.



















Tell Us What You Think!