-
OpCT-001 is the first iPSC-derived investigational cell therapy to be clinically tested for treating primary photoreceptor diseases
-
Phase 1/2a trial, CLARICO, is a first-in-human, multisite, 2-part interventional trial to evaluate the safety, tolerability, and the effect on clinical outcomes of OpCT-001
-
Primary photoreceptor diseases are a subgroup of inherited retinal disorders that affect the structure and function of the photoreceptor cells in the retina, leading to irreversible vision loss in children and adults
Future Medical Products LLC Announces the Launch of PBME Topical™ (Pure Bone Marrow Exosomes)
July 24, 2025, St. Louis, Missouri — Future Medical Products LLC is pleased to announce the launch of PBME Topical (“Pure Bone Marrow Exosomes”). PBME Topical is a cosmetic product in compliance with the Modernization of Cosmetics Regulation Act of 2022 (MoCRA).
PBME Topical consists of isolated exosomes from mesenchymal stem cells, the most studied cell in science. These exosomes are composed of phospholipid bilayers with transmembrane proteins and receptors, integrated growth factors, in addition to micro-RNA and messenger-RNA. Exosomes are naturally found in every part of the body, but the source of the exosomes intended for aesthetics, as well as regenerative medicine, is critical. Bone marrow exosomes are considered by some to be the purest form of stem cell therapy without the cell. [Read more…]
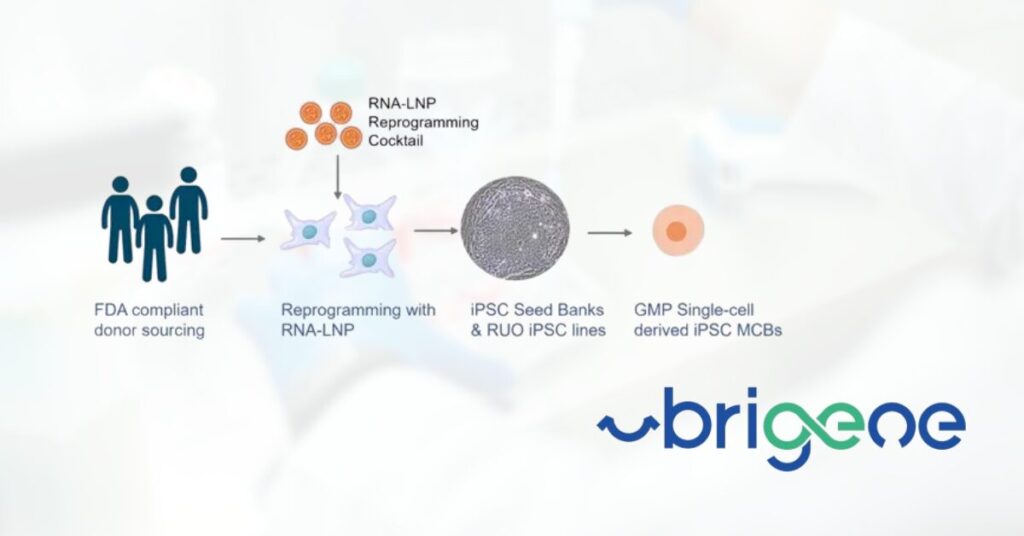
uBriGene Launches Clinical iPSC Banks to Accelerate Regenerative Medicine and Cell Therapy Development
ROCKVILLE, MD, June 16, 2025 – uBriGene Biosciences, a leading cell and gene therapy CDMO, is proud to announce the launch of its fully characterized, ready-to-use induced pluripotent stem cell (iPSC) banks. Developed using uBriGene’s proprietary RNA-LNP reprogramming technology, these iPSC banks are now available to support research, translational development, and clinical applications worldwide. [Read more…]
Vita Therapeutics and I Peace announced a development program to generate iPS- derived differentiated cells for transplant therapy with an initial focus on FSHD (Facioscapulohumeral Muscular Dystrophy) using universal iPS cells
Palo Alto, CA, July 10, 2025 – Leading GMP cell CDMO I Peace, Inc., specializing in induced pluripotent stem cells (iPSCs) and iPSC-derived cell therapies, announced that the company has agreed with Vita Therapeutics (Vita) to jointly develop universal iPS cells that will be used for cell transplantation therapy.
GMP-grade iPS cells will be modified with proprietary, licensed gene editing technology to make a base platform hypoimmune line where Class I and II HLA genes that determine rejection have been depleted. These hypoimmune lines can be further customized in I Peace cGMP facilities. [Read more…]
Ciloa secures €6.5 million government funding from France 2030 ‘Innovation in biotherapies and biomanufacturing’ to develop drug candidate for treatment of obesity and type 2 diabetes
- « Previous Page
- 1
- …
- 9
- 10
- 11
- 12
- 13
- …
- 116
- Next Page »