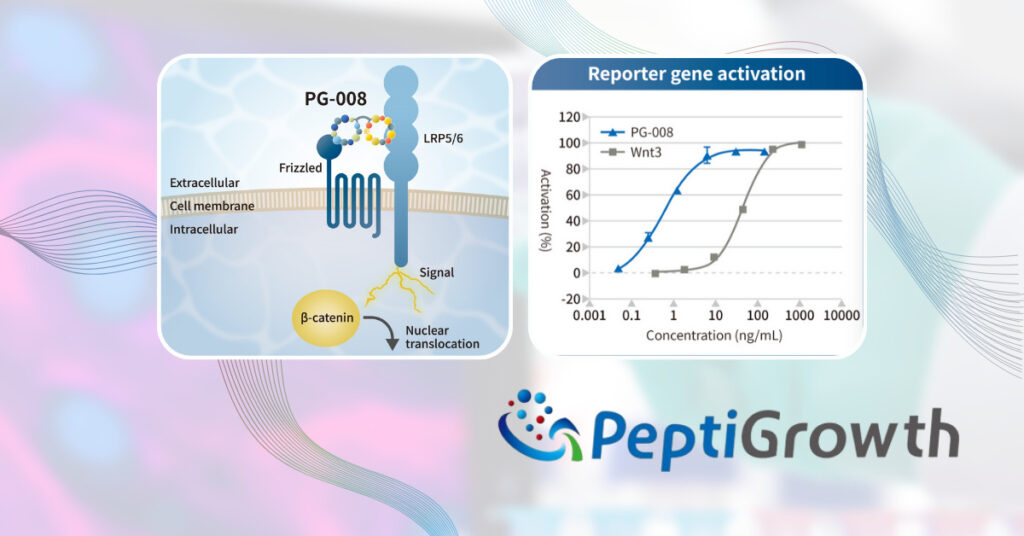
PeptiGrowth Inc. (Headquarters: Chiyoda-ku, Tokyo, President: Jiro Sugimoto) has successfully developed a novel synthetic peptide called “Wnt3a alternative peptide (β-catenin pathway agonist)” which has equivalent function to recombinant Wnt3a as well as GSK-3β inhibitors, such as CHIR99021(CHIR)). This product will be available in the middle of November 2023. [Read more…]
VCCT Inc., Leading the Field of Retinal Regeneration through iPS Cells, Raises 600M Japanese yen ($4M USD) through Third-Party Allotment
Cumulative Funds Raised Amount to Approximately 1.7 billion yen (Approximately $11.3 million USD)
KOBE, Japan — VCCT Inc. (President: Masayo Takahashi) is pleased to announce the completion of a private equity allotment of approximately 600 million yen ($4 million USD). The allotment involved new shares to Sysmex Corporation (President: Kaoru Asano), DAI-DAN CO., LTD. (Representative Director, President: Ichiro Fujisawa), and YASKAWA Electric Corporation (Representative Director, President: Masahiro Ogawa). VCCT will continue to accelerate the development of retinal regenerative medicine together with these partners. [Read more…]
Ajinomoto Co. to Acquire Gene Therapy CDMO Forge Biologics for an Astounding $620 Million
- Acquisition extends global capabilities in AAV and plasmid gene therapy manufacturing for Ajinomoto, Co. Inc., diversifying and strengthening their current contract development and manufacturing services
- Positions Forge Biologics for accelerated long-term growth supporting gene and cell therapy clients worldwide
Century Therapeutics and FUJIFILM CDI Announce Licenses for the Development and Commercialization of iPSC-Derived Cell Therapies in Autoimmune and Inflammatory Diseases
PHILADELPHIA and MADISON, Wis. — Century Therapeutics, Inc. (NASDAQ: IPSC), an innovative clinical-stage biotechnology company developing induced pluripotent stem cell (iPSC)-derived cell therapies in immune-oncology, and FUJIFILM Cellular Dynamics, Inc., a leading global developer and manufacturer of human iPSCs and iPSC-derived cells, today announce a new worldwide license agreement and expansion of existing licenses for the development and commercialization of cell therapies derived from iPSCs for the treatment of autoimmune and inflammatory diseases, including type 1 diabetes, multiple sclerosis, lupus, and rheumatoid arthritis. [Read more…]
Atsena Therapeutics Receives FDA RMAT Designation for ATSN-101 Gene Therapy
RMAT designation recognizes the potential of ATSN-101 to address unmet medical needs for patients with LCA1
ATSN-101 has demonstrated clinically meaningful improvements in vision at the highest dose with no drug-related serious adverse events 6 months post-treatment in ongoing Phase I/II clinical trial
- « Previous Page
- 1
- …
- 5
- 6
- 7
- 8
- 9
- …
- 97
- Next Page »