The cord blood banking market is an interesting market, as its rate of growth has slowed over the past couple years. When the market is assessed by the number of cord blood units stored, cord tissue units banked, and number of units transplanted, there are regions that are experiencing growth and regions in decline. In particular, cord blood markets are experiencing strong growth within Asia, are relatively flat within North America, and are in decline within Europe. Additionally, cord blood banks within Europe have been some of the most proactive banks to expand into other areas of stem cell storage. [Read more…]
Actinium Receives Orphan Drug Designation From FDA for Iomab-B to Condition Patients for HSCT
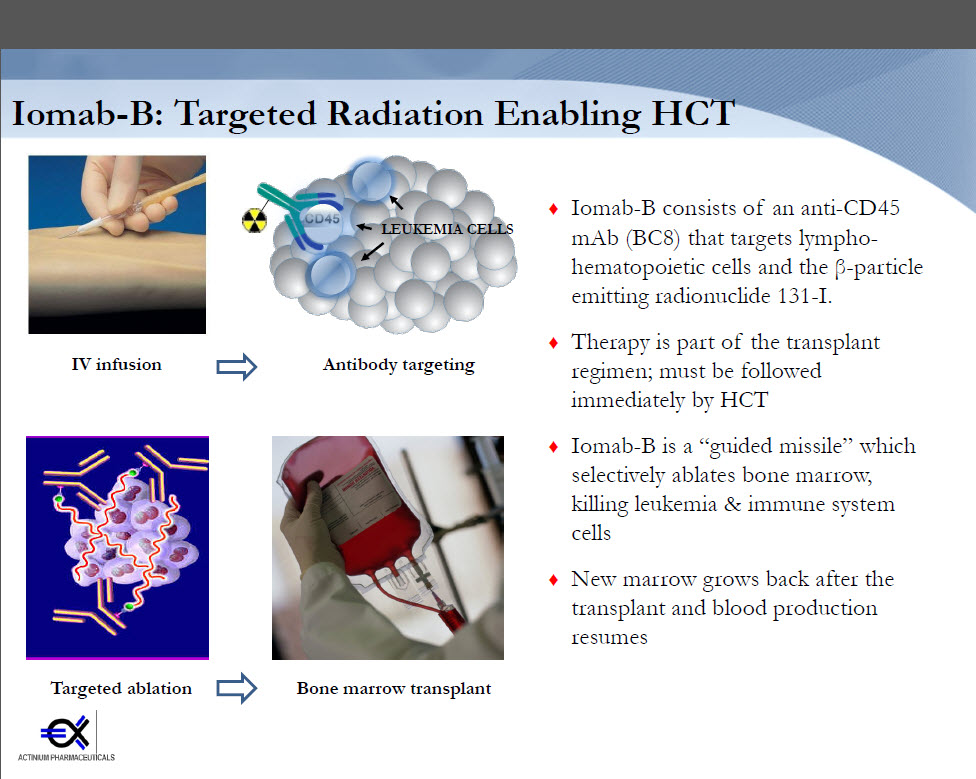
In major news released today, Actinium Pharmaceuticals announced that the FDA has “granted orphan drug designation for Iomab-B, a radioimmuno-therapeutic that conditions relapsed and refractory Acute Myeloid Leukemia (AML) patients for a hematopoietic stem cell transplant (HSCT).”
The drug is key to condition patients undergoing a hematopoietic stem cell transplant, making it a significant announcement for the stem cell community.
To learn more, read the full press release below, printed with permission from Janet Vasquez, Managing Director/Founder of JV Public Relations NY and media contact for Actinium Pharmaceuticals, Inc. [Read more…]
HemaCare Expands Its Offering of Stem Cell-Enriched Peripheral Blood
LOS ANGELES—HemaCare Corporation (OTCBB:HEMA) a leader in cell and tissue collection, processing and cell therapy solutions, announced that it has expanded its offering of stem cell rich peripheral blood through the addition of a new protocol.
This type of blood, referred to among researchers as “mobilized peripheral blood,” is collected following a procedure that induces the bone marrow to produce more white cells into the periphery. These white cells can then be collected with a routine leukapheresis and provided to researchers within 24 hours.
Where is the Stem Cell Industry Headed? (We Interviewed 6 Executives to Find Out)
Without question, some of BioInformant’s most popular content has been our exclusive interviews with stem cell executives and key opinion leaders (KOL). Highlighted below are six of our most popular interviews. Enjoy! [Read more…]
CardioCell Expands Chronic Heart Failure Phase IIa Clinical Trial to MedStar Washington Hospital Center
In news released today, CardioCell, LLC, announced that a fifth treatment center has been chosen for participation in its Phase II clinical trial that will investigate the use of allogeneic mesenchymal bone marrow cells (aMBMC) in the treatment of non-ischemic heart failure (Study #NCT02467387). Now recruiting patients for this fifth location, the clinical study launched June 2014 and has an estimated end date of April 2017.
To learn more about the announcement, see the full press release below, printed with permission from Marcie Frank, Stemedica’s Vice President, Investor Relations. [Read more…]
- « Previous Page
- 1
- …
- 266
- 267
- 268
- 269
- 270
- …
- 313
- Next Page »