|
|
Can T cells be an effective approach to cancer treatment? CAR T-cell therapy, as this cutting-edge treatment is called, uses a patient’s own immune system to help fight cancers in the body. The result is a natural treatment that proves effective against a variety of blood cancers and may have hope for other cancers in the future as well.
T-cells are the workhorses of our immune system and they play a key role in directing the immune response and killing cells infected by pathogens.
Using T Cells For Cancer Treatment
In this article:
- CAR-T Cell Therapy Treatments for Treatment of Cancer
- What Causes Cancer?
- How Does the Immune System Work – and What Are Its Limits?
- What Is Immunotherapy?
- Are There Different Types of Immunotherapy?
- What Are T Cells?
- What Is CAR-T Cell Therapy?
- How Does CAR-T Cell Therapy Work?
- Who Is CAR-T Cell Therapy Right For?
- Why Doesn’t CAR-T Cell Therapy Work for Solid Tumors?
- Are There Any Risks?
- What Are the Benefits of CAR-T?
- How Effective Is CAR-T Treatment?
- Progress in CAR-T Cell Therapy Development
CAR-T Cell Therapy Treatments for Treatment of Cancer
Cancer is an age-old enemy of the human race, and while we have shown great promise in fighting back these past few decades, it still takes a huge number of lives every year. According to the National Cancer Institute, approximately 1.8 million cases of cancer are diagnosed each year, and more than 600,000 people will die of it. According to the Leukemia and Lymphoma Society, a person gets diagnosed with blood cancer every 3 minutes. Luckily, a new therapy has emerged, leveraging the power of the T cell.
Currently, the following CAR-T therapies have been approved by FDA, including:
- Tisagenlecleucel-T (Kymriah)
- Axicabtagene ciloleucel (Yescarta)
- Brexucabtagene autoleucel (Tecartus)
- Lisocabtagene maraleucel (Breyanzi)
- Idecabtagene vicleucel (Abecma)
- Ciltacabtagene autoleucel (Carvykte)
In addition, two CAR-T cell products have been approved in China: Relma cabtagene autoleucel (relma-cel) and Axicabtagene ciloleucel (Yikaida). The two earliest approvals were Kymriah, which was approved by FDA in August 2017, and Yescarta, which was approved by FDA in October 2017.
This means blood cancer sufferers who have not found other options may finally have a solution. While the treatments are still highly experimental, their rarity is decreasing and effectiveness increasing.
In order to understand this emerging form of immunotherapy, however, it is first critical to understand a little bit about cancer. It’s important to know what causes it and how the body typically fights disease. From there, readers will benefit from understanding exactly what a T cell is and how physicians can put it to work. For those considering the treatment, understand whom it’s for and what risks are involved. Patients should also know the benefits and long-term effects.
Learn how re-engineered bodily cells may become the best soldiers in the fight against the second-biggest killer in the United States today.
What Causes Cancer?
While cancer is widely regarded as a terrifying killer, it is not widely understood on a scientific level. In order to see how CAR T cell therapy works, however, you need to have a firm grasp of what cancer is and what it means for the body.
Essentially, cancer is a disease that involves the uncontrollable growth of cells. Instead of growing at a typical rate and dying off as normal cells are programmed to do, cancer cells divide quickly and don’t die when they should. Worse, cancer can spread from one system to another – known as metastasis – through infiltration methods of which other cells are incapable.
These gene mutations may result from genetic anomalies with which the patient is born. They often run the family, or as a result of the surrounding environment. Common causes of cancer, known as carcinogens, include smoking, chemicals, obesity, unbalanced hormones, radiation, lack of exercise, and chronic inflammation.
So how does cancer occur? As a result of gene mutations. These mutations cause the body’s programming code, DNA, to direct rapid and uncontrollable growth instead of sedate reproduction. It also fails to send signals to stop growing and dividing at the proper time. With each division of unhealthy cancer cells, these incorrect instructions get magnified. It continues to do so until the body can no longer overcome the damage.
Thanks to screening and treatment options that improve all the time, life expectancy and survival rates have increased substantially. However, the medical community, patients, and loved ones remain unsatisfied with the stubborn nature of some cancer cases.
Immunotherapy, luckily, stands poised to increase the odds in favor of patients even further.
How Does the Immune System Work – and What Are Its Limits?
The immune system is one of the most powerful forces in medicine. It keeps us safe from viruses, bacteria, and toxins. It consists of many organs that control immunity. These special cells that carry out the immune system’s orders, and chemicals and substances designed to fight “invasions” when they arise. These special cells and compounds are mobile, capable of traveling through the body to the site of infection.
These invaders are known as antigens. They are foreign substances that the body doesn’t recognize as normal or healthy. When it targets substances or germs that don’t belong, the immune system sends cells to the site to attack them. These cells develop antibodies, specialized proteins designed to neutralize the threat.
Here’s the kicker: Where the immune system encounters such substances, it builds an immunity. This means it keeps the recipe for those antibodies on hand and thus “remembers” how to fight the invaders later. (That’s the basis of vaccines, which offer a controlled dose of a disease, helping the body develop a resistance to that particular disease agent without risking full exposure.)
So why does cancer pose such a distinct threat to the body still? Because the invaders, in this case, are from the body itself. That means the immune system often has a hard time recognizing cancer cells as something it ought to attack. Moreover, the cells themselves can, in many cases, disarm the immune system by sending out certain chemical signals.
Luckily, by reprogramming some of the body’s natural immune mechanisms, researchers and physicians can help patients’ bodies overcome these obstacles.
What Is Immunotherapy?

Again, the immune system is incredibly powerful. While manmade treatments provide an invaluable addition to the disease-fighting toolkit, the body’s own strategies of war still number among the most effective of all. It should come as no surprise, therefore, that cutting-edge immunotherapy treatments have swept the world in the last several years.
In a nutshell, immunotherapy is a type of medicine that uses the body’s own defensive devices against cancer and other diseases. Unlike the inherent system, however, which may not prove strong enough to repel cancer’s attack, immunotherapy responses are much more intense. To create them, researchers help immune cells recognize cancer more effectively and deal with it more harshly.
Are There Different Types of Immunotherapy?
The good news continues: There exist more than one type of immunotherapy. In designing several options, researchers have helped to bolster the cancer-fighting toolkit further. When one approach doesn’t work for a particular patient or cancer type, others remain available. According to the American Cancer Society, the most common types of immunotherapy include:
Monoclonal Antibodies
Immune system proteins are designed to fight infection. While the body’s naturally manufactured versions often get the job done, they don’t always – hence the existence of cancer in the first place.
Monoclonal antibodies manufactured in a lab often have a better chance of having an intended effect, because they receive very specific instructions from researchers about which part of the cancer cell to attack. This makes this much more effective than the body’s more generalized approach.
Immune Checkpoint Inhibitors
Again, one of the ways cancer cells accomplish their uncontrolled growth is to release chemicals that tell the immune system not to attack them. These are called “checkpoints.” While their function is to help immune cells distinguish between healthy and unhealthy cells before initiating an immune response, this makes immune cells vulnerable to trickery. If those checkpoints protect cancer cells, the body loses one of its most powerful defenses.
That’s where immune checkpoint inhibitors come in. As the American Cancer Society also explains, these drugs take the brakes off the immune system, so that it attacks without hesitation.
Cancer Vaccines
Remember, vaccines give the body a controlled dose of disease. That’s so it can learn to fight the disease without risking normal levels of exposure – which the body might not prove capable of overcoming. Although we typically think of vaccines as a preventative measure, they are also effective as a treatment. By introducing low doses of the vaccine in question, the body can make the antibodies needed to overcome the targeted disease.
Non-Specific Immunotherapies
Rather than targeting a specific disease as the others do, this type of immunotherapy generally strengthens the immune system, providing it with better all-around defenses against cancer attack. In some cases, it is enough simply to bolster the system, at which point it can handle cancer cells on its own.
Adoptive Cell Immunotherapy
This is where scientists take a patient’s immune cells and treat them in a lab to make them better at their job. They reprogram them, in effect, to help them target cancer much more effectively. By changing their genetic code, they help cells recognize cancer cells that might previously have hidden from immune system cells.
CAR-T cell therapy falls under this category.
What Are T Cells?
T cells are one of the body’s most important soldiers in the fight against invaders of all sorts, including cancer. It is responsible for recognizing pathogens and creating highly tailored immune responses to deal with them.
T cells are born in bone marrow. While young, they are called T stem cells. From there, they travel to the thymus to mature. They receive exposure to a hormone called thymosin, as well as several other factors designed to turn them into the killer cells the body needs to deal with pathogens, toxins, and cancers. Any T cells that develop abnormally get killed during the maturation process. That includes T cells in danger of attacking the body rather than hazarding against it. This helps limit autoimmune disorders, in which the immune system itself sees normal tissue as invaders they have to fight.
There exist many purposes for T cells, the functions of which researchers do not entirely understand. Scientists do know that they produce cytokines, a type of protein that helps prompt a further immune response. Because T cells are so important, they’re often used as a determining factor in HIV/AIDS patients, as well as others with immunodeficiency disorders.
Because T cells are so critical in the fight against cancer, it’s a real problem when those cancer cells manage to fool them. But there’s a solution to that.
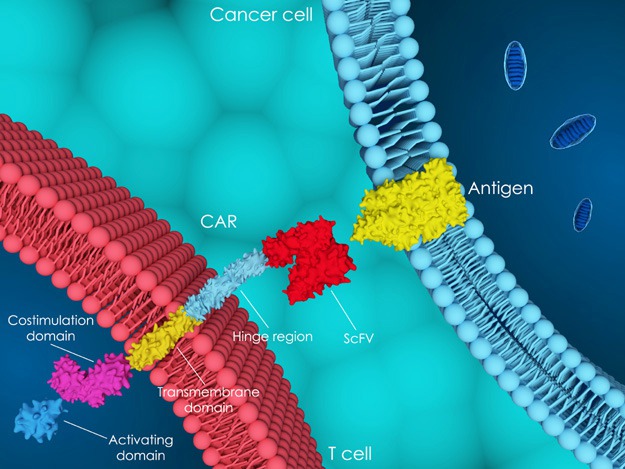
What Is CAR-T Cell Therapy?
CAR-T cell therapy is a type of adoptive cell immunotherapy. That means it relies on researchers to tweak the genetic code so that cancer cells can no longer hide from the immune system through checkpoint inhibitors and other means that convince the body they are normal cells rather than invaders.
The therapy works by giving T cells the ability to bind to cancer cells. These cells initiate an immune response against them, killing them. Because the genetic reprogramming is so highly specialized, there is little danger that the cells will harm healthy cells in the body. However, that is not a given; see risks below.
How Does CAR-T Cell Therapy Work?
In order to administer CAR-T cell therapy to patients, physicians must first have their T cells on hand. That means drawing blood, done during a simple draw from a vein in the patient’s arm. The tube flows into an apheresis machine, a device capable of separating out different components in the blood. The machine isolates T cells, then gives the remaining blood back to the patient.
Once physicians have the patients T cells, they activate them using certain antibodies. This prepares them for changes to the genetic code, delivered via viral vector. A virus receives a genetic code and then introduced into T cells, dumps its genetic load into the cells. This, therefore, gives them the ability to recognize and attack cancer.
Specifically, the virus conveys the genetic instructions for manufacturing chimeric antigen receptors – hence CAR. It could also generate special receptors that allow the T cell to recognize the form of cancer attacking the patient. The T cell now has the tools needed to seek and destroy cancers that might formerly have hid from the immune system.
Once the T cells have that ability, they are cultured in a lab to produce a high number of them. When physicians deemed cells have reached appropriate levels, they reintroduce them back into the patient via infusion, but not before testing for sterility to ensure doctors do not inadvertently introduce other infections.
Once inside the body, T cells will both attack cancer and continue to multiply.
Who Is CAR-T Cell Therapy Right For?
Most obviously, CAR-T cell therapy is for patients with blood disorders. Specifically, these include:
Childhood Acute Lymphoblastic Leukemia
Also known as ALL, this of leukemia affects 75 percent of children diagnosed with leukemia. Today’s treatments for this type of cancer are extremely effective these days, with 90 percent of them living 5 years or more. However, children and young adults still die of it at a rate of about 600 per year. That’s because the current treatments can’t necessarily stop patients from relapsing. In the case of a relapse, the patient usually only has weeks or months to live.
The good news? In 52 of 63 patients, CAR T cell therapy caused the cancer to go back into remission, reports Cancer.Net. This is an incredible accomplishment and represents a major leap forward in the treatment of childhood leukemia. The therapy responsible, called tisagenlecleucel, has received FDA approval.
Non-Hodgkin Lymphoma
This type of blood cancer, of which diffuse large B-cell lymphoma (DLBCL) is most common, is very aggressive. While chemotherapy and other treatments often prove effective, at times they don’t. They sometimes get worse during treatment or they may appear effective only for the patient to relapse afterward.
CAR-T cell therapy may prove effective in these cases. Clinical trials show positive results, with 43 percent of patients going into remission, according to the Cancer.Net source. The FDA has also approved a CAR-T cell treatment for this, known as axicabtagene ciloleucel.
Multiple Myeloma
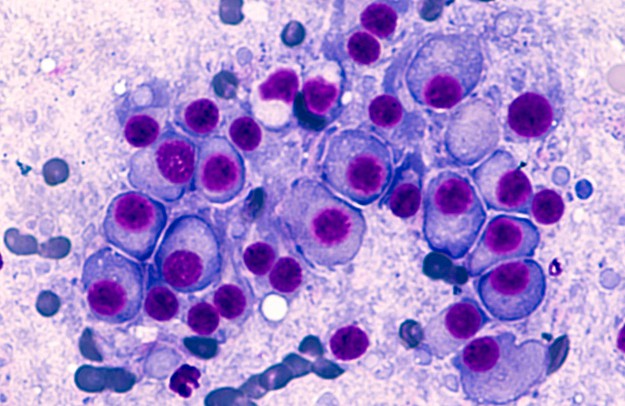
Plasma cells play a critical role in protecting the body against disease and invaders. Multiple myeloma, which damages these cells, can prove life-threatening. Roughly half of the patients will die within 5 years of their diagnosis.
While the FDA has not yet approved a treatment for this type of blood cancer, clinical trials show positive results – perhaps the most positive yet, with 33 of 35 patients going into remission within 2 months of treatment, says the same source.
Unfortunately, while the procedures are hopeful indeed, they are not yet widely available. Those that have received approval are typically only open to children and adults whose other treatments have failed to work. In some cases, failed multiple times. Those interested in the treatments can get the right information from physicians who have the knowledge to tailor information to their specific cases.
Why Doesn’t CAR-T Cell Therapy Work for Solid Tumors?
Unfortunately, CAR-T cell therapy doesn’t appear to be very effective against solid tumors – at least not yet. Solid tumor are those that form a stationary mass in any of the body’s organs or tissues. That’s because solid tumors are typically a result of many mutations in the genetic code, so it’s harder for researchers to target what’s causing them and how to fight those causes.
Like some blood cancers, solid tumors are also adept at using the body’s own immune system to hide and protect themselves. This makes fighting them extra difficult.
That’s not to say researchers won’t someday develop a form of this treatment that can do more than the current capability. Researchers just have to figure out what drives the tumor’s growth, at which point they’ll have a lot more information about how to reprogram T cells to counteract that. But for now, the FDA and medical community have put their focus on blood cancers.
Are There Any Risks?
Unfortunately, CAR-T cell therapy does carry a risk of severe side effects. One of the most dangerous is CRS, or cytokine release syndrome. Recall that cytokines are proteins that stimulate an immune response. While that is beneficial in many cases, there exists the danger that cytokines – much like cancer – will get out of control. When they do, they can attack healthy cells and tissue, which makes patients very sick and may even prove life-threatening.
- Flu-like symptoms
- Fever
- Neurological issues
- Heart trouble
- Organ failure
- Anorexia
- Clotting disorders
… and more.
Because of this, hospitals require that staff trained in the management of CRS remain on hand throughout the entire procedure. Also because of the possibility of complications – and the need to wait for T cells to reach the right proportions to fight cancer – the procedure may take up to several weeks from start to finish. It also must happen in a hospital setting after T cell reintroduction.
It’s also possible for cytokines to attack healthy B cells, which is especially dangerous given these cells are partially responsible for preventing infection.
Another risk, less particular to patients and more generally to the process as a whole, is its extreme complexity. It takes weeks for T cells to receive the new genetic information, grow to certain levels, pass sterility tests, and become ready for re-injection. All of these has to happen before the patient’s hospital stay. This has raised questions in the medical community of whether the process can really work on a mass scale.
As of now, however, researchers have not given up.
What Are the Benefits of CAR-T?
The biggest benefit of this therapy is that CAR-T cells are designed to work forever. Through giving the cells new genetic code, physicians effectively give patients the ability to fight that particular cancer for the rest of time. T cells will then replicate as normal, distributing the virally-introduced information throughout the body.
Therefore, when relapses occur, the body already has the tools on hand to fight the disease. It can initiate an immune response against that specific cancer without having to wait, and the cancer can never hide again, disguise itself as healthy tissue, or disrupt the immune system’s ability to attack it.
Because of this, CAR T cell therapy and others like it are known as “living drugs,” which will remain in the body for the rest of the patient’s life.
How Effective Is CAR-T Treatment?
The short answer is that CAR-T cell therapy has shown some extremely promising results. Patients who didn’t respond to radiation, chemotherapy, and surgery a few years ago – and therefore had essentially no chances of recovery – have responded to this treatment. That’s not to say it works for everyone, because that’s true for no treatment.
For now, studies are confined to an extremely small sample of the population. As already discussed, that sample has typically proven resistant to other treatments. However, because of the high success rates, researchers are searching for ways to make it more widely available, even for those who have not yet demonstrated resistance to more typical approaches. Once this happens, researchers will have a better idea of how often it works across the board, as opposed to only in these tough cases.
Progress in CAR-T Cell Therapy Development
Even more promising is the rapid rate at which development of the treatment has progressed. CAR-T cell therapy only reached commercialization in 2017 with the approval of Kymriah. Since then, a total of six CAR-T cell therapies have been approved by the U.S. FDA—Kymriah, Yescarta, Tecartus, Breyanzi, Abecma, and Carvykte—and two CAR-T cell products have been approved in China, relma-cel and Yikaida.
This rate of progress is extremely hopeful and indicates that scientists have a lot more to discover when it comes to this groundbreaking therapy.
For patients and loved ones curious about whether or not they have access to the procedure, the best approach is to bring up it up during consultation with the physician presiding over your treatment. Patients may qualify for clinical trials or the physician can help expedite whatever other treatments the patient might need to try first.
Interested in learning more about CAR-T cell therapy and other cutting-edge medical approaches? We invite you to join our community.
Would you be interested in a cancer treatment that uses your body’s own T cells? Share your thoughts in comments.
Up Next: CAR-T Companies: The Meteoric Rise Of Cellular Immunotherapies



















Tell Us What You Think!