|
|
Since the discovery of induced pluripotent stem cell (iPSC) technology 18 years ago, significant progress has been made in stem cell biology and regenerative medicine. New pathological mechanisms have been identified, new drugs identified by iPSC screens are in the pipeline, and the first clinical trials employing human iPSC-derived cell types have been initiated.
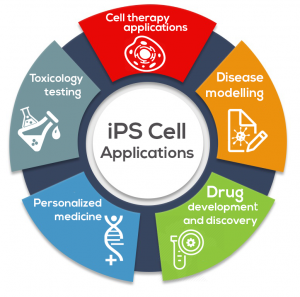
Today, methods of commercializing iPSCs include:
Cell Therapy: iPSCs are being explored in a diverse range of cell therapy applications for the purpose of reversing injury or disease.
Disease Modelling: By generating iPSCs from patients with disorders of interest and differentiating them into disease-specific cells, iPSCs can effectively create disease models “in a dish.”
Drug Development and Discovery: iPSCs have the potential to transform drug discovery by providing physiologically relevant cells for compound identification, target validation, compound screening, and tool discovery.
Personalized Medicine: The use of techniques such as CRISPR enable precise, directed creation of knock-outs and knock-ins (including single base changes) in many cell types. Pairing iPSCs with genome editing technologies is adding a new dimension to personalized medicine.
Toxicology Testing: iPSCs can be used for toxicology screening, which is the use of stem cells or their derivatives (tissue-specific cells) to assess the safety of compounds or drugs within living cells.
Proliferating Applications for iPSCs
Other applications of iPSCs include their use as research products, as well as their integration into 3D bioprinting, tissue engineering, and increasingly, clean meat production. Plus, technology allowing for the mass-production and differentiation of iPSCs in industrial-scale bioreactors is advancing at breakneck speed, with companies like TreeFrog Therapeutics leading the charge.
In recent years, iPSC-derived cells have increasingly been used within preclinical testing and early stage-stage trials. The first clinical trial using iPSCs started in 2008, and today, that number has surpassed 100 worldwide. Most of these clinical trials do not involve the transplant of iPSCs into humans, but rather, the creation and evaluation of iPSC lines for clinical purposes. Within these trials, iPSC lines are created from specific patient populations to determine if these cell lines could be a good model for a disease of interest.
However, there are companies forging forward with clinical trials involving iPSC-derived cell therapeutics, such as Cynata Therapeutics (and its partner Fujifilm), Healios K.K. (in collaboration with Sumitomo Dainippon Pharma), and Fate Therapeutics.
Other companies involved with the commercialization of iPSC-derived cell therapeutics include:
- Allele Biotechnology and Pharmaceuticals is developing a diabetes drug created from iPSC-derived pancreatic beta cells.
- Aspen Neuroscience is combining stem cell biology and genomics to provide the world’s first autologous induced pluripotent stem cell (iPSC)-derived neuron replacement therapy for Parkinson disease.
- Avery Therapeutics and I Peace, Inc., are collaborating to advance an iPSC-derived cell therapeutic for heart failure. I Peace is generating and supplying GMP-grade iPSCs, while Avery Therapeutics is using them to manufacture its MyCardia™ product.
- Bayer acquired iPSC cell therapy company BlueRock Therapeutics in August 2019. Since May 2021, BlueRock Therapeutics, Fujifilm Cellular Dynamics, and Opsis Therapeutics have had an R&D alliance to develop allogeneic iPSC-derived cell therapies for ocular diseases.
- BlueRock Therapeutics, a subsidiary of Bayer since August 2019, develops iPSC-derived cell therapies to target Parkinson’s disease, heart failure, and ocular diseases.
- Bone Therapeutics has partnered with the U.S. company Implant Therapeutics to develop allogeneic, iPSC-derived MSCs.
- Brooklyn Immuno Therapeutics is developing a set of mesenchymal stem cell (MSC) products, derived from iPSCs, to which it also intends to apply its gene editing technology.
- CellOrigin Biotech (Hangzhou) Co, Ltd – CellOrigin Biotech and Qilu Pharmaceutical are co-developing allogeneic iPSC-derived chimeric antigen receptor macrophages (CAR-iMAC) to assist in the treatment of cancer.
- Cellectis – Cellectis has a partnership with Cytovia Therapeutics for the production of gene-edited iNK cells (NK cells derived from iPSCs). Cytovia uses Cellectis’ TALEN gene-editing technology to produce its iNK cells while minimizing the risk of off-target effects.
- Century Therapeutics was created in July 2019 by Versant Ventures and Fujifilm to develop iPSC-derived adaptive and innate immune effector cell therapies.
- Citius Pharmaceuticals uses iPSCs from a single-donor dermal fibroblast to create iPSC-derived MSCs (i-MSCs). It has completed the development of an i-MSC Accession Cell Bank (ACB) and is testing and expanding these cells to create an allogeneic cGMP i-MSC Master Cell Bank.
- Clade Therapeutics is developing immune-cloaked pluripotent stem cell derived therapies, including iPSC-derived cell therapies
- Cynata Therapeutics manufacturers iPSC-derived MSCs using its proprietary Cymerus™ technology. In partnership with FUJIFILM Corporation, it is clinically testing these cells for the treatment of graft-versus-host disease (GvHD). It is also conducting trials for the treatment of critical limb ischemia (CLI), osteoarthritis (OA), and respiratory failure/distress, including ARDS.
- CytoMed Therapeutics Pte Ltd (“CytoMed)” is engaged in clinical translation of its human iPSC derived gamma delta natural killer T cell technology. This technology enables the generation of a novel, hybrid 2-in-1 cell type, which combines receptors of both NK and T cells in an allogeneic setting to recognise a broad spectrum of cancers, including both solid and hematological malignancies. CytoMed Therapeutics (NASDAQ : GDTC) listed on NASDAQ in April of 2023.
- Cytovia Therapeutics is a biopharma company developing allogeneic “off-the-shelf” gene-edited iNK and CAR (Chimeric Antigen Receptor)-iNK cells derived from iPSCs.
- Edigene, Inc. – Edigene and and Neukio Biotherapeutics are developing allogenic iPSC-derived NK cell therapies through a joint R&D collaboration.
- Editas Medicine (Nasdaq: EDIT), a genome editing company, is developing engineered iPSC-derived natural killer cells (iNKs) for the treatment of cancer.
- Eterna Therapeutics – Eterna’s gene-edited iPS cell therapeutic candidates promote enhanced immune responses, including effecting T-cell mediated immunity and promoting multilineage differentiation and hematopoietic regeneration in vivo. Eterna entered into a sponsored research agreement with MD Anderson in November 2022 to further this program.
- Exacis Biotherapeutics is a development-stage immuno-oncology company that is developing NK cells from iPSCs (ExaNK™ cells) engineered using mRNA gene-editing technology to resist rejection by the patient’s immune system.
- Fate Therapeutics is developing iPSC-derived NK and CAR-T cells for the treatment of cancer and immune disorders.
- FUJIFILM Cellular Dynamics, Inc. (FCDI) is investing in a $21M cGMP production facility to support its internal cell therapeutics pipeline, as well as serve as a CDMO for iPS cell products.
- Greenstone Bio – Greenstone Bio is a start-up company headquartered in Stanford Research Park in Palo Alto, California. The companies couples artificial intelligence (AI) with patient-derived iPSC technology to accelerate the drug discovery process. It has extensive knowledge and expertise in generating patient-derived iPSCs, as well as the largest patient-derived iPSC Biobank. It also offers cell lines to academic collaborators at no cost.
- Heartseed Inc. is a Japanese biotech company that is developing iPSC-derived cardiomyocytes (HS-001) for the treatment of heart failure. The company is positioned to initiate a phase 1/2 study of this investigational cell therapy in Japan in the second half of 2021.
- Healios K.K., in collaboration with Sumitomo Dainippon Pharma, is undertaking a clinical trial using allogeneic iPSC-derived retinal cells to treat age-related macular degeneration.
- Hebecell is commercializing allogeneic “off-the-shelf” iPSC-derived NK cell therapeutics. The company uses a unique feeder-free 3D-iPSC spheroid platform to produce NK cells. This efficient, scalable and reproducible method could allow nearly unlimited production of iPSC-derived NK cells for the treatment of cancer and other diseases.
- Hopstem Biotechnology is one of the first iPSC cell therapy companies in China and a market leader in iPSC-derived clinical-grade cell products. In June 2021, it partnered with Neurophth Biotechnology to co-develop an iPSC-derived cell therapy for the treatment of ocular diseases. Hopstem has a proprietary neural differentiation platform, as well as a patented iPSC reprogramming method and GMP manufactory and quality systems.
- Implant Therapeutics is engineering iPSC-MSC cells containing FailSafe™ and induced Allogeneic Cell Tolerance (iACT Stealth Cell™) technologies. These iPSC MSC cells are hypo-immunogenic and can be used as ex-vivo gene therapy vehicles.
- I Peace Inc. and Avery Therapeutics are collaborating to advance an iPSC-derived cell therapeutic for heart failure. I Peace is generating GMP-grade iPSCs, while Avery Therapeutics is using them to manufacture its MyCardia™ product. I Peace is able to mass production clinical-grade iPSC lines simultaneously in a single room using a miniaturized plate and robotic technology, and its facility is equipped with a fully-closed automated iPSC manufacturing system that meets the safety standards of the U.S. FDA and Japanese PMDA.
- iPSirius SAS is an immuno-oncology firm focused on developing IPVAC 1.0, a novel therapeutic cancer vacci
ne. Leveraging the antigenic overlap between iPSCs and cancer stem cells (CSCs), iPSirius aims to combat relapse and metastatic progression in cancer patients. - IPS HEART – IPS HEART is a privately owned cell therapy company that has developed programs to reprogram iPSCs into skeletal muscle and cardiac muscle for the treatment of disease. Specifically, IPS HEART uses small molecules to reprogram iPSCs into new, functional heart muscle for the treatment of heart failure, Duchenne Cardiomyopathy and Danon Cardiomyopathy, and new skeletal muscle for the treatment of Duchenne muscular dystrophy.
- Jacobio Pharmaceuticals has a strategic investment with US-based Hebecell to develop iPSC-derived NK cells for the treatment of cancer and other diseases.
- Keio University won approval from the the Japanese government in February 2018 for an iPSC trial that involves the treatment of patients with spinal cord injuries (led by Professor Hideyuki Okano).
- Kyoto University Hospital, in partnership with the Center for iPS Cell Research and Application (CiRA), is performing a physician-led study of iPSC-derived dopaminergic progenitors in patients with Parkinson’s disease.
- Laverock Therapeutics – Laverock is to utilizing Gene Editing induced Gene Silencing (GEiGS®) technology, and its associated computational platform, to engineer iPSC-derived cell therapies that have improved efficacy, safety, and accessibility.
- Neurophth Biotechnology Ltd. is a gene therapy company specializing in AAV-mediated gene therapies for the treatment of ocular diseases. In June 2021, it partnered with Hopstem Biotechnology to develop an iPSC-derived candidate cell product for an agreed upon retinal degenerative disorder.
- Notch Therapeutics is developing iPSC-derived T cells for treating hematological malignancies, in partnership with Allogene Therapeutics.
- Novo Nordisk signed a co-development agreement with Heartseed in mid-2021 that grants it exclusive rights to develop, manufacture, and commercialize HS-001 globally, excluding Japan where Heartseed retained exclusive rights to develop HS-001. HS-001 is an investigational therapy comprised of purified iPSC-derived ventricular cardiomyocytes for the treatment of heart failure.
- Osaka University grafted a sheet of iPS-derived corneal cells into the cornea of a patient with limbal stem cell deficiency, a condition in which corneal stem cells are lost.
- Pluristyx and panCELLa – In October 2022, Pluristyx merged with panCELLa. Today, the combined company has a goal is be the global leader in clinical-grade, genetically modified, “off-the-shelf” iPSCs with the lowest barrier to entry for cell therapy development.
- REPROCELL recently launched a “Personal iPS service” in Japan to prepare and store an individual’s iPSCs for the treatment of future illness or injury. Individual’s iPSCs are created from mature cells in their urine or dental pulp, using RNA reprogramming technology. The iPSCs are then stored at two locations in Japan and the U.S.
- RIKEN administered the world’s first iPSC-derived cell therapeutic into a human patient in 2014 when it transplanted an autologous iPSC-RPE cell sheet into a patient with AMD.
- RheinCell Therapeutics GmbH is a developer and manufacturer of GMP-compliant human iPSCs derived from HLA-homozygous, allogeneic umbilical cord blood. In January 2021, the company received GMP certification and Manufacturing Authorization within the EU.
- RxCell Inc. has manufactured a Master Cell Bank of iPSCs for allogenic therapy, including IND activities for retinal degenerative disorders. It has also developed a novel hypoimmunogenic strategy and made universal iPSC lines that can be used to manufacture differentiated cells for allogenic therapy.
- Ryne Biotechnology, Inc. – Ryne Bio is a therapeutics company leveraging iPSC technology to develop a platform of off-the-shelf neuron replacement therapies for neurological disorders. It has received a $4 million Clinical Stage Research Program (CLIN1) grant from CIRM to enable it to advance its lead candidate RNDP-001, an iPSC-derived dopamine neuron progenitor for the treatment of inherited and idiopathic forms of Parkinson’s disease, through submission of an IND.
- Sana Biotechnology – Sana Biotechnology is developing hypoimmune-modified allogeneic (“donor”) iPSCs to evade immune response and rejection without immunosuppression. It is currently testing this cell technology in non-human primates (monkeys).
- SCG Cell Therapy Pte Ltd (“SCG”) has acquired the rights to human iPSC technology, from the Agency for Science, Technology and Research (“A*STAR”)’s Accelerate Technologies Pte Ltd (“A*ccelerate”). SCG is using this iPSC technology to to expand its cell therapy product portfolio and develop off-the-shelf NK cell therapies.
- SCM Lifescience, a South Korean stem cell therapy developer, licensed exclusive rights within Korea for the development, approval, production, and sale of a diabetic cell therapy being developed by Allele Biotechnology and Pharmaceuticals. The $750K deal was signed in July 2021.
- Semma Therapeutics, which was acquired by Vertex Pharmaceuticals for $950 million in late 2019, is developing a treatment for Type 1 diabetes. This treatment consists of cells derived from iPSCs that behave like pancreatic cells.
- Shoreline Biosciences is a biotech company that is developing allogeneic “off-the-shelf” natural killer (NK) and macrophage cellular immunotherapies derived from iPSCs for cancer and other serious diseases.
- Stemson Therapeutics has been developing a therapy for hair loss involving generation of de novo hair follicles.
- TreeFrog Therapeutics has a 13,000 sq ft facility in France for the development and scale-up of its cell therapy manufacturing process that leverages human iPSCs. It plans to develop its own iPSC-derived therapies and support co-development programs.
- The U.S. NIH is undertaking the first U.S. clinical trial of an iPSC-derived therapeutic. Its Phase I/IIa clinical trial will involve 12 patients with advanced-stage geographic atrophy of the eye.
- Vita Therapeutics, a Cambrian Biopharma affiliate, is developing iPSC-derived therapeutics. VTA-100 is an autologous, genetically engineered iPSC-derived therapeutic for the treatment of limb-girdle muscular dystrophy, while VTA-200 is a genetically engineered iPSC-derived hypoimmunogenic treatment for muscular dystrophy.
Of course, there are numerous physician-led studies employing human iPSC-derived cell types that have been undertaken as well, including ones by Keio University, Kyoto University Hospital, Osaka University, and RIKEN.
Within the research products realm, the number of market competitors has surged in recent years, although the following industry stalwarts continue to defend their position as iPSC market leaders: Fujifilm Cellular Dynamics, Inc.(USA/Japan), REPROCELL (Japan), Evotec (Europe), Ncardia (Europe), and Axol Bioscience (Europe).
Finally, the world’s largest research supply companies are also commercializing a plethora of iPSC-related products. Examples of these market leaders include Lonza, BD Biosciences, Thermo Fisher Scientific, Merck, Takara Bio, and others.



Tell Us What You Think!