SHANGHAI, Oct. 28, 2019 — CARsgen Therapeutics Co. Ltd., a clinical-stage biopharmaceutical company today announced that the United States Food and Drug Administration (FDA) has granted Regenerative Medicine Advanced Therapy (RMAT) designation to its investigational CT053 CAR-T cell therapy. CT053 is a fully human anti-BCMA (B Cell Maturation Antigen) autologous chimeric antigen receptor (CAR) T Cell therapy for the treatment of relapsed and/or refractory multiple myeloma (rrMM). [Read more…]
Stem Cell News
BioInformant's coverage of stem cell news includes iPS cells, mesenchymal stem cells, hematopoietic stem cells, neural stem cells and more.
As the first and only market research firm to specialize in stem cell news, BioInformant research has been cited by the Wall Street Journal, Xconomy, AABB, and Vogue Magazine.
Serving Fortune 500 leaders that include GE Healthcare, Pfizer, and Goldman Sachs, BioInformant is your global leader in stem cell industry news.
Platelet BioGenesis Bags $56M to Develop Human iPSC-Derived Platelets as Nuclear Countermeasure
Highlights:
- Platelet BioGenesis has the world’s first technology to generate clinical-grade human platelets from induced pluripotent stem cells (iPSCs).
- Platelet BioGenesis has been awarded $56 Million Contract from the Biomedical Advanced Research and Development Authority (BARDA) to develop donor-independent platelets as a treatment for radiological and nuclear exposure.
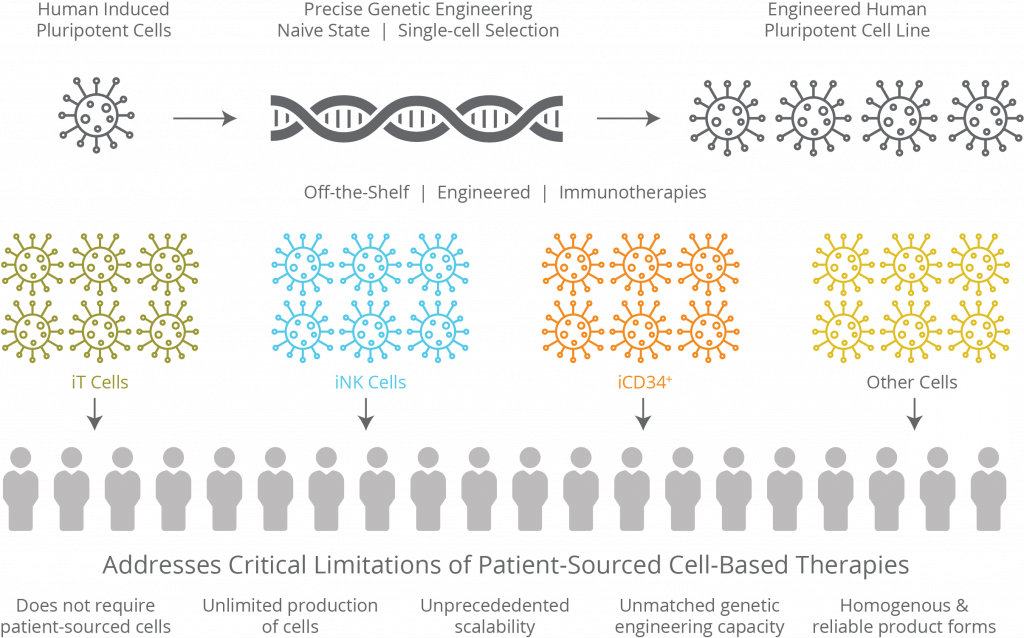
Fate Therapeutics Announces the Opening of its cGMP Manufacturing Facility Dedicated to iPSC-derived Cell Therapies
State-of-the-Art Facility Designed to use Clonal Master iPSC Lines as Renewable Cell Source for Manufacture of Off-the-Shelf Product Pipeline
 SAN DIEGO, Sept. 30, 2019 — Fate Therapeutics, Inc. (FATE), a clinical-stage biopharmaceutical company dedicated to the development of programmed cellular immunotherapies for cancer and immune disorders, announced today that the Company has opened its current Good Manufacturing Process (cGMP) compliant manufacturing facility for the clinical production of its off-the-shelf natural killer (NK) cell and chimeric antigen receptor (CAR) T-cell product candidates. [Read more…]
SAN DIEGO, Sept. 30, 2019 — Fate Therapeutics, Inc. (FATE), a clinical-stage biopharmaceutical company dedicated to the development of programmed cellular immunotherapies for cancer and immune disorders, announced today that the Company has opened its current Good Manufacturing Process (cGMP) compliant manufacturing facility for the clinical production of its off-the-shelf natural killer (NK) cell and chimeric antigen receptor (CAR) T-cell product candidates. [Read more…]
Positive Study Results of Phase IIa Clinical Trial Using IV Administered MSCs for Ischemic Stroke Published in Peer-Reviewed Stroke Journal
SAN DIEGO, Calif. – September 30, 2019 – Results from a study sponsored by Stemedica Cell Technologies, Inc., a global biotechnology company that uses allogeneic stem cells for ischemic conditions, form the basis for a peer-reviewed paper published in Stroke entitled “Phase I/II Study of Safety and Preliminary Efficacy of Intravenous Allogeneic Mesenchymal Stem Cells in Chronic Stroke.” Co-authors include Michael L. Levy, MD, PhD, John R. Crawford, MD, Nabil Dib, MD, Lev Verkh, PhD, Nikolai Tankovich, MD, PhD and Steven C. Cramer, MD. [Read more…]
Anemocyte to expand its plasmid manufacturing capabilities
 A new state-of-the-art facility dedicated to plasmid manufacturing is now operational in Gerenzano, Italy. The new site will couple with the existing GMP facility active in the Cell and Gene (C&G) therapy space. With 800m2 (8,000 square feet) of cleanrooms, the two facilities of Anemocyte, the first Biotech Manufacturing Organisation (BMO), will address manufacturing needs of Somatic Cells, Non-Viral Modified Cells, Vesicles and Plasmids for Viral Vectors. [Read more…]
A new state-of-the-art facility dedicated to plasmid manufacturing is now operational in Gerenzano, Italy. The new site will couple with the existing GMP facility active in the Cell and Gene (C&G) therapy space. With 800m2 (8,000 square feet) of cleanrooms, the two facilities of Anemocyte, the first Biotech Manufacturing Organisation (BMO), will address manufacturing needs of Somatic Cells, Non-Viral Modified Cells, Vesicles and Plasmids for Viral Vectors. [Read more…]
- « Previous Page
- 1
- …
- 81
- 82
- 83
- 84
- 85
- …
- 123
- Next Page »