First ever known successful regeneration of full-thickness skin and hair; Company poised to initiate human trial in the third quarter of 2017; Management to host conference call Thursday, June 8th at 4:30pm ET
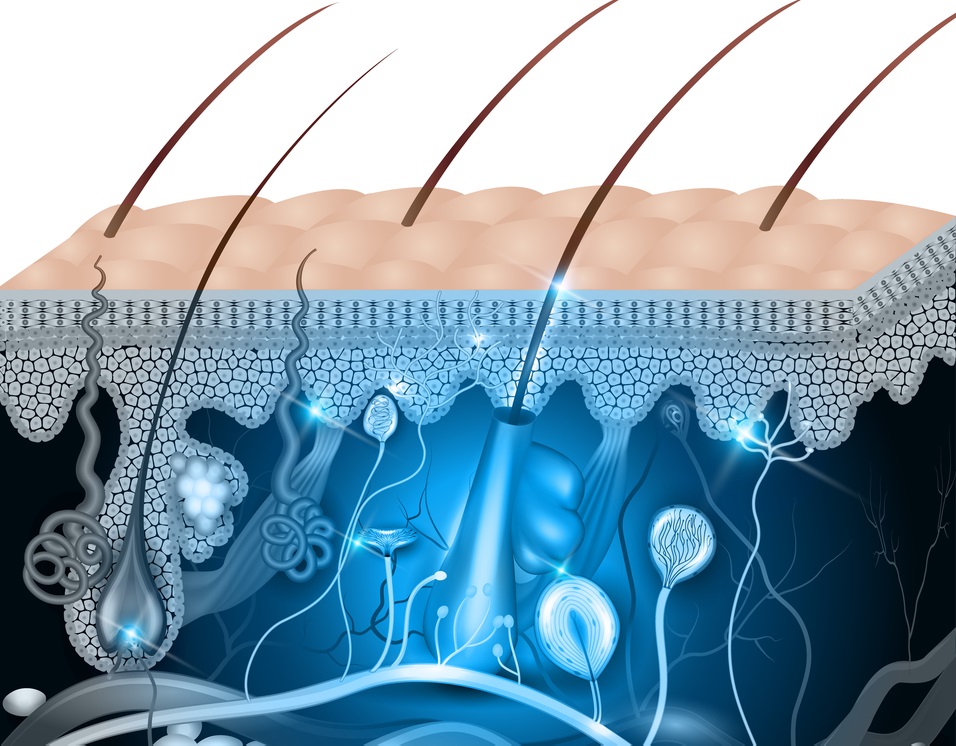
 SALT LAKE CITY, UT, 06/08/17 — PolarityTE™, Inc. (NASDAQ: COOL) today announced pre-clinical results demonstrating that the Company’s lead product, SkinTE™, regenerated full-thickness, organized skin and hair follicles in third degree burn wounds. The findings represent the first known successful regeneration of skin and hair in full-thickness swine wound models, the standard animal model for human skin. The Company expects to initiate a human clinical trial evaluating the autologous homologous SkinTE™ construct in the third quarter of 2017. [Read more…]
SALT LAKE CITY, UT, 06/08/17 — PolarityTE™, Inc. (NASDAQ: COOL) today announced pre-clinical results demonstrating that the Company’s lead product, SkinTE™, regenerated full-thickness, organized skin and hair follicles in third degree burn wounds. The findings represent the first known successful regeneration of skin and hair in full-thickness swine wound models, the standard animal model for human skin. The Company expects to initiate a human clinical trial evaluating the autologous homologous SkinTE™ construct in the third quarter of 2017. [Read more…]



 Leiden and Oss, The Netherlands, May 30th, 2017 – Pluriomics and Pivot Park Screening Centre announced a collaboration to improve Pluriomics’ cardiovascular drug discovery service offerings and to expand Pivot Park Screening Centre’s knowledge about new and innovative assay technologies used to accelerate drug discoveries. Giving Pluriomics access to the Screening Centre’s ultra High Throughput Screening (uHTS) capability, this collaboration will serve Pluriomics’ mission which is to contribute to the efficiency of the drug discovery and development process by reducing the development length, costs and late stage attrition of novel drug candidates.
Leiden and Oss, The Netherlands, May 30th, 2017 – Pluriomics and Pivot Park Screening Centre announced a collaboration to improve Pluriomics’ cardiovascular drug discovery service offerings and to expand Pivot Park Screening Centre’s knowledge about new and innovative assay technologies used to accelerate drug discoveries. Giving Pluriomics access to the Screening Centre’s ultra High Throughput Screening (uHTS) capability, this collaboration will serve Pluriomics’ mission which is to contribute to the efficiency of the drug discovery and development process by reducing the development length, costs and late stage attrition of novel drug candidates.