 SAN CARLOS, Calif., Aug. 22, 2018 — In a Viewpoint paper published online today in Circulation Research, the Phase III CardiAMP Heart Failure Trial (CardiAMP-HF Trial) studying the investigational CardiAMP stem cell therapy from BioCardia®, Inc. (OTC: BCDA) was highlighted as an example of personalized medicine, with promising results from the first 10 patients in the roll-in cohort reported. The company believes this to be the first outcomes data published from any pivotal trial of a stem cell therapy for heart failure. The paper is authored by Peter Johnston, M.D., Eric Duckers, M.D., Ph.D., Amish Raval, M.D., Thomas Cook, Ph.D. and Carl Pepine, M.D.
SAN CARLOS, Calif., Aug. 22, 2018 — In a Viewpoint paper published online today in Circulation Research, the Phase III CardiAMP Heart Failure Trial (CardiAMP-HF Trial) studying the investigational CardiAMP stem cell therapy from BioCardia®, Inc. (OTC: BCDA) was highlighted as an example of personalized medicine, with promising results from the first 10 patients in the roll-in cohort reported. The company believes this to be the first outcomes data published from any pivotal trial of a stem cell therapy for heart failure. The paper is authored by Peter Johnston, M.D., Eric Duckers, M.D., Ph.D., Amish Raval, M.D., Thomas Cook, Ph.D. and Carl Pepine, M.D.
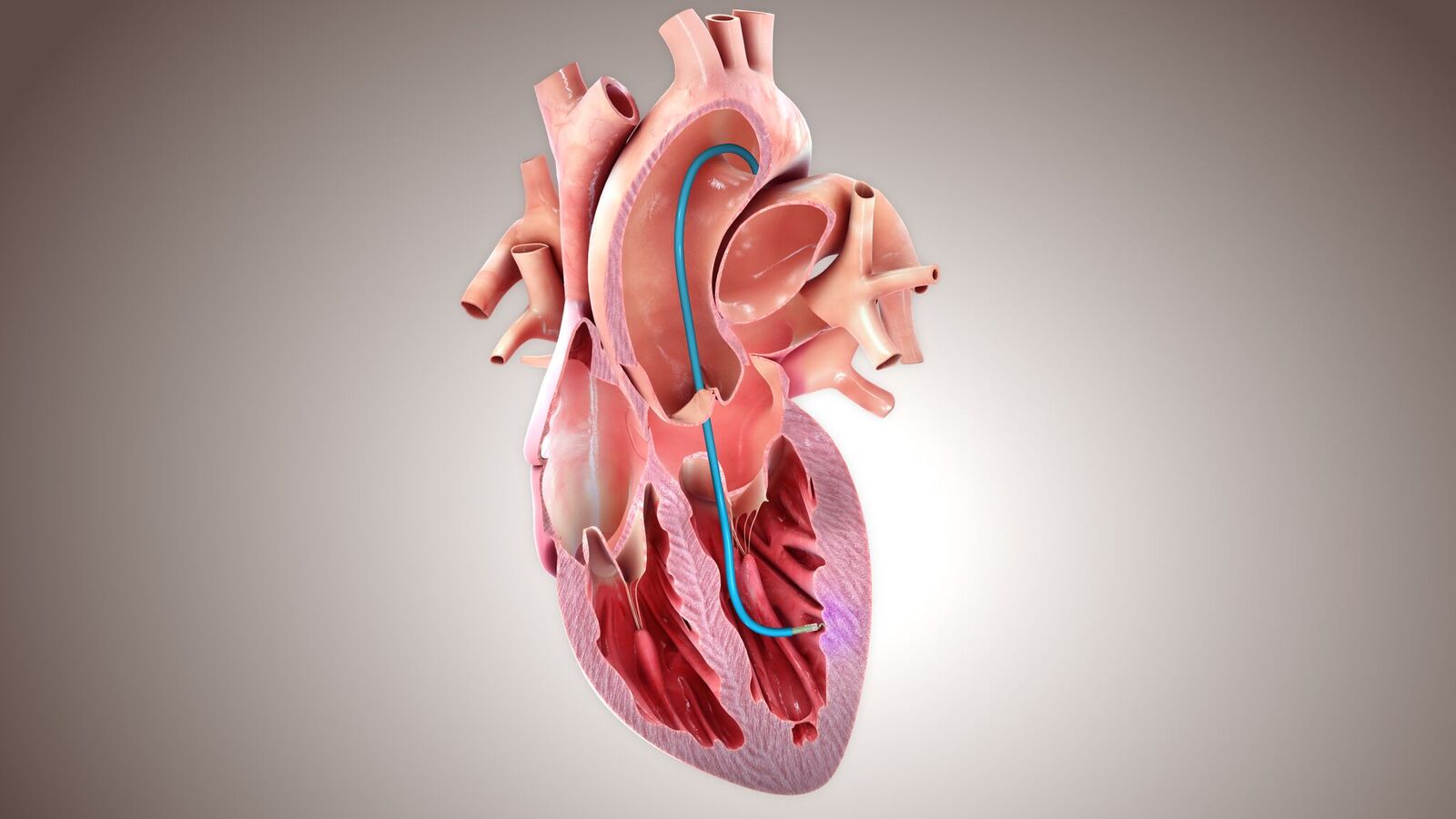
The investigational CardiAMP therapy uses the patient’s own (autologous) bone marrow cells to potentially stimulate the body’s natural healing response through a minimally-invasive, catheter-based procedure. BioCardia is a leader in the development of comprehensive solutions for cardiovascular regenerative therapies.
In the article entitled, “Not All Stem Cells Are Created Equal: The Case for Prospective Assessment of Stem Cell Potency in the CardiAMP Heart Failure Trial,” Peter Johnston, M.D. and his co-authors state, “Using a ‘personalized’ medicine approach (i.e. tailoring treatment to patient characteristics to optimize benefit), we propose to optimize cardiac cell therapy efficacy by prospectively selecting patients most likely to benefit using a cell potency assay to assess inherent regenerative capacity.”
 In the article, they write that the varying properties and potency of bone marrow-derived stem cells across patients are recognized, but little focus has been put on the regenerative capacity of the patients themselves in determining outcomes. However, they cite several studies that have retroactively identified cell characteristics and other markers associated with improved outcomes, such as higher counts of B-cells and CXCR4+ bone marrow mononuclear cells, and lower levels of pre-procedural VEGF, erythropoietin and CXCL 10/IP10, among others.
In the article, they write that the varying properties and potency of bone marrow-derived stem cells across patients are recognized, but little focus has been put on the regenerative capacity of the patients themselves in determining outcomes. However, they cite several studies that have retroactively identified cell characteristics and other markers associated with improved outcomes, such as higher counts of B-cells and CXCR4+ bone marrow mononuclear cells, and lower levels of pre-procedural VEGF, erythropoietin and CXCL 10/IP10, among others.
They point out that the pivotal CardiAMP-HF Trial is the first to incorporate a cell potency analysis and score in the screening criteria. During screening, a bone marrow aspiration is drawn to measure surface marker expression and functional cell properties to identify likely responders, who are then randomized and scheduled for treatment.
The paper reports for the first time on the results from the first 10 patients in the open-label Roll-In Phase of the CardiAMP-HF Trial treated with CardiAMP therapy. Treated patients showed improvements in Six Minute Walk Distance (+47.8m ± 19.6, 20.5% relative improvement; p=0.01), improvements in New York Heart Association Heart Failure Class (40% of patients improving one class, p=0.037), and a positive trend in MLHFQ Score (-10.2 ± 7.9, 31% relative improvement, p=0.21) at six months when compared to their baseline values. The magnitude of these improvements is greater than those seen in the trial’s predecessor, the double blind, Phase II Transendocardial Autologous Cell Therapy in Heart Failure Trial, which demonstrated statistical significance in functional capacity as measured by six-minute walk and quality of life as measured by the Minnesota Living with Heart Failure Questionnaire.
The authors conclude that, “Delivery on the promise of regenerative therapy will require the development of novel strategies leveraging knowledge gained in prior trials to prospectively identify patients likely to respond to therapy to optimize therapeutic potency through evidence-based cell characterization, preparation, and delivery. CardiAMP-HF is a first step in this direction. We believe that trials offering more specific, patient-centered and ‘personalized’ approaches to regenerative medicine will generate the robust and effective therapies needed to address CHF (congestive heart failure) and other conditions.”
Following online publication today, the article is scheduled to be published in print in Circulation Research on September 28.
The 260-patient CardiAMP-HF Trial is designated by the Center for Biologics Evaluation and Review within the U.S. Food and Drug Administration as a pivotal study, intended to provide the primary support for the safety and effectiveness evaluation of the therapy for the purposes of a marketing application. The trial is currently enrolling at 18 world-class U.S. centers, and up to 40 U.S. centers may ultimately participate in the study. The CardiAMP-HF Trial is supported by nondilutive trial reimbursement support from the Center for Medicare and Medicaid Services and nondilutive grant support from the Maryland Stem Cell Research Fund.
About BioCardia
BioCardia, Inc., headquartered in San Carlos, California, is developing regenerative biologic therapies to treat cardiovascular disease. CardiAMP and CardiALLO cell therapies are the Company’s biotherapeutic product candidates in clinical development. The Company’s current products include the Helix™ transendocardial delivery system and the Morph® steerable guide and sheath catheter portfolio. BioCardia also partners with other biotherapeutic companies to provide its Helix systems and clinical support to their programs studying therapies for the treatment of heart failure, chronic myocardial ischemia and acute myocardial infarction.
Forward Looking Statements
This press release contains forward-looking statements that are subject to many risks and uncertainties. Forward-looking statements include, among other things, references to the enrollment in our clinical trials, the intended outcomes of our trials, the efficacy and safety of our products and therapies, statements regarding our intentions, beliefs, projections, outlook, analyses or current expectations. Such risks and uncertainties include, among others, the inherent uncertainties associated with developing new products or technologies, regulatory approvals, unexpected expenditures, the ability to raise the additional funding needed to continue to pursue BioCardia’s business and product development plans and overall market conditions. We may find it difficult to enroll patients in our clinical trials due to many factors, some of which are outside of our control. Slower than targeted enrollment could delay completion of our clinical trials and delay or prevent development of our therapeutic candidates. These forward-looking statements are made as of the date of this press release, and BioCardia assumes no obligation to update the forward-looking statements.
We may use terms such as “believes,” “estimates,” “anticipates,” “expects,” “plans,” “intends,” “may,” “could,” “might,” “will,” “should,” “approximately” or other words that convey the uncertainty of future events or outcomes to identify these forward-looking statements. Although we believe that we have a reasonable basis for each forward-looking statement contained herein, we caution you that forward-looking statements are not guarantees of future performance and that our actual results may differ materially from the forward-looking statements contained in this press release. As a result of these factors, we cannot assure you that the forward-looking statements in this press release will prove to be accurate. Additional factors that could materially affect actual results can be found in BioCardia’s Annual Report on Form 10-K filed with the Securities and Exchange Commission on March 16, 2018, including those under the caption titled “Risk Factors.” BioCardia expressly disclaims any intent or obligation to update these forward-looking statements, except as required by law.
Investor Contact:
David McClung, Chief Financial Officer
Email: [email protected]
Phone: 650-226-0120
Media Contact:
David Schull | Alex Xenakis
Email: [email protected] | [email protected]
Phone: 212-845-4271 | 212-845-4226
SOURCE BioCardia, Inc.
http://www.biocardia.com



















Tell Us What You Think!