|
|
PALO ALTO, Calif., Nov. 1, 2023 — Leading GMP cell CDMO I Peace, Inc., specializing in induced pluripotent stem cells (iPSCs) and iPSC-derived cell therapies based in Palo Alto, CA, and Heartseed Inc. (“Heartseed”), a Tokyo-based biotechnology company developing iPSC-derived cardiomyocytes for heart failure, have collaborated to generate cardiomyocytes using multiple donor-derived GMP grade iPS cell lines by I Peace, and Heartseed’s proprietary cardiomyocytes differentiation and purification methods. Heartseed has successfully produced high-purity cardiomyocytes consistently from all I Peace manufactured iPS cell lines used in this study. This achievement marks a significant step forward in the realization of autologous cardiac regenerative medicine with I Peace’s personal iPS cell banking service (My Peace), to manufacture and store iPS cells, and induce them into cardiomyocytes for use in autologous cell therapy.
Significance of stable production of high-purity cardiomyocytes derived from iPS cells
A major advantage of medical treatment using autologous iPS cells is that immunosuppressive agents are not necessary because no immune reaction occurs when cells are transplanted. However, the differentiation efficiency of iPS cells into target cells varies, and to efficiently produce cardiomyocytes from different donor-derived iPS cells, it is necessary to optimize the method of cardiomyocyte production for each iPS cell. This has been a hurdle to the realization of medical treatment using autologous iPS cells.
Background of the study
I Peace has developed a technology to simultaneously generate iPS cells from multiple donors for their personalized iPS cell banking service (My Peace) and has been able to produce numerous clinical grade iPS cells affordably and reliably.
Heartseed is developing HS-001 (open chest administration) and HS-005 (catheter administration), which are microtissues of highly purified cardiomyocytes (cardiomyocyte spheroids) generated from allogeneic iPS cells and is accumulating know-how on the properties of cardiomyocytes and efficient methods of inducing cardiomyocyte differentiation.
In this context, I Peace and Heartseed have been investigating the production of high-purity cardiomyocytes using multiple donor-derived iPS cell lines produced by I Peace, using Heartseed’s original method of cardiomyocyte differentiation and purification.
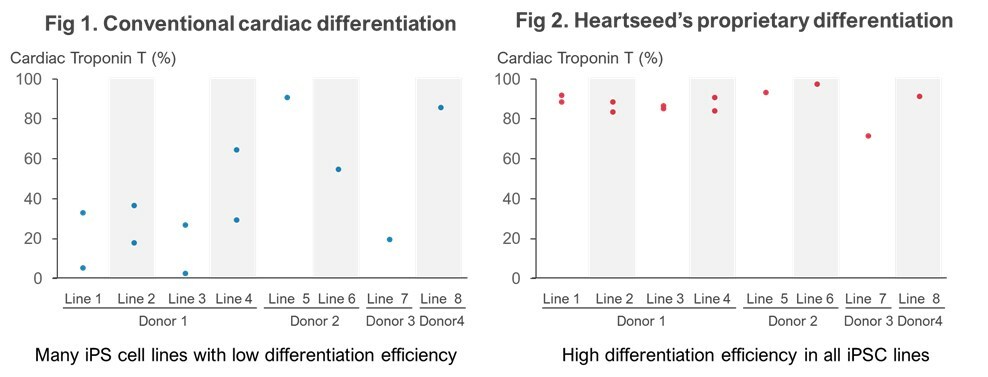
When a conventional cardiomyocyte differentiation method was applied, low differentiation efficiency was observed in many iPS cell lines (Figure 1). On the other hand, by applying Heartseed’s original cardiomyocyte differentiation method, high differentiation efficiency into cardiomyocytes was observed from all eight iPS cell lines from four different donors (Figure 2). Furthermore, by applying Heartseed’s proprietary purification method “Metabolic Selection” to the obtained cardiomyocytes, nearly 99% purity was achieved in all eight lines, making them suitable for medical applications.
I Peace will continue to work together with Heartseed towards establishing regenerative medicine for heart failures using patients’ own iPS cells. I Peace will also collaborate with various institutions in other fields.
About Heartseed
Heartseed Inc. was founded in 2015 to develop and commercialize cardiac remuscularization therapy developed by Keiichi Fukuda and his group at the Department of Cardiology, Keio University, Tokyo, Japan. Heartseed has proprietary technologies throughout the entire manufacturing process of the cardiomyocyte-cell product, including purification, cell delivery, and iPSC production.
Heartseed announced the global collaboration and license agreement with Novo Nordisk A/S for HS-001 in June 2021. Heartseed received “Minister of Science and Technology Policy Award” at Japan Venture Awards 2021 and “Ministry of Education, Culture, Sports, Science and Technology Award” at Academic Startups 2021, and “Most Promising Pipelines Awards (iPSC)” at Asia Pacific Cell & Gene Therapy Excellence Awards 2022. For more information, visit heartseed.jp, LinkedIn, and YouTube.
About I Peace
I Peace Inc. (https://www.ipeace.com), a GMP cell development and contract manufacturing company specializing in iPS cells and iPS cell-derived cell therapy, sells cGMP iPS cells and provides contract manufacturing services for medical cells globally. The company was founded in 2015 by Koji Tanabe, a graduate of Professor Shinya Yamanaka’s lab at Kyoto University and the second author of the paper that reported the world’s first successful establishment of human iPS cells. Tanabe, who has been involved in iPS cell research since the beginning of its development, is committed to making iPS cells accessible to all people through I Peace. I Peace’s unique technology enables the parallel production of multiple donor-derived iPS cells without contamination concerns and makes it possible to provide a large number of iPS cells at a reasonable cost. I Peace supports drug discovery and cell therapy development through the use of the iPS cell and other cell products that meet PMDA and FDA standards as high-quality cell products for pharmaceutical and cell therapy companies, and also promotes the production of iPS cells for individuals so that each and every person in the world can prepare for the future by possessing his or her own iPS cells. In addition to supporting pharmaceutical and cell therapy companies so that cell therapy will be within the reach of patients as soon as possible, I Peace aims to spread regenerative cell therapy as soon as possible by establishing iPS cell banking services for individuals.
Heartseed Inc.
CEO: Keiichi Fukuda
Established: 2015
Head office: Tokyo, Japan
Website: https://heartseed.jp/en/company.html
Phone/E-mail: 381-3-6380-1068 [email protected]
I Peace, Inc.
Founder & CEO: Koji Tanabe
Established: 2015
Headquarters: Palo Alto, California, U.S.A.
Subsidiary in Japan: I Peace, Ltd., (Kyoto)
iPS cell manufacturing base: Peace Engine Kyoto, Kyoto, Japan
Website: https://www.ipeace.com

Tell Us What You Think!