|
|
Since the discovery of induced pluripotent stem cell (iPSC) technology 16 years ago, significant progress has been made in stem cell biology and regenerative medicine. New pathological mechanisms have been identified, new drugs identified by iPSC screens are in the pipeline, and the first clinical trials employing human iPSC-derived cell types have been initiated.
Commercializing iPS Cells
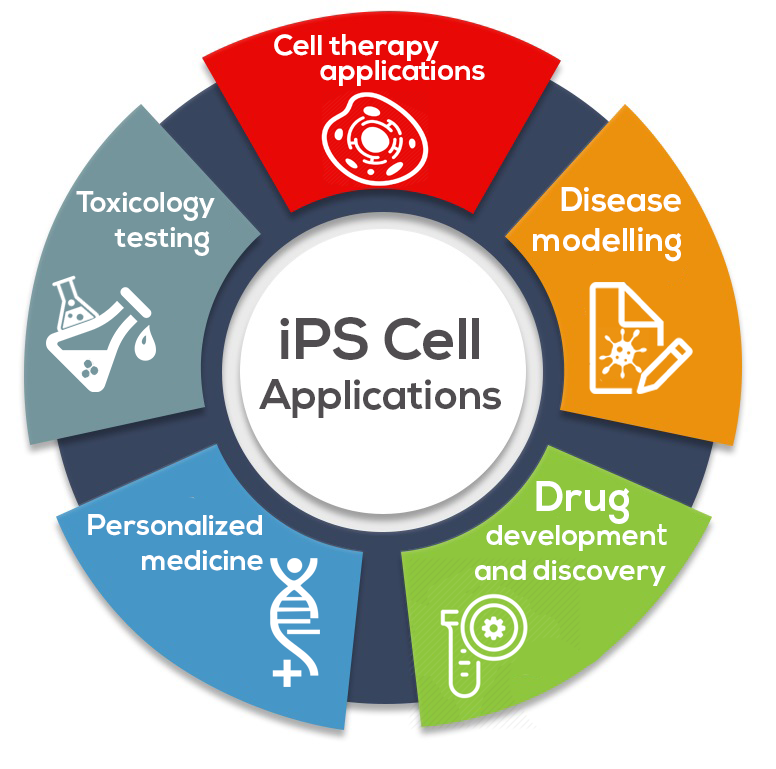
Today, methods of commercializing iPSCs include:
1. Cell Therapy
iPSCs are being explored in a diverse range of cell therapy applications for the purpose of reversing injury or disease.
There are many examples of this, but a leading one is Cynata Therapeutic’s historic trial utilizing its iPSC-derived product, CYP-004, in 440 patients with osteoarthritis (OA). This was a landmark trial because it was the world’s first Phase 3 trial to administer an iPSC-derived cellular therapeutic to human patients and the largest one to date.
2. Disease Modelling
By generating iPSCs from patients with disorders of interest and differentiating them into disease-specific cells, iPSCs can effectively create disease models “in a dish.”
3. Drug Development and Discovery
iPSCs have the potential to transform drug discovery by providing physiologically relevant cells for compound identification, target validation, compound screening, and tool discovery.
4. Personalized Medicine
The use of techniques such as CRISPR enable precise, directed creation of knock-outs and knock-ins (including single base changes) in many cell types. Pairing iPSCs with genome editing technologies is adding a new dimension to personalized medicine.
5. Toxicology Testing
iPSCs can be used for toxicology screening, which is the use of stem cells or their derivatives (tissue-specific cells) to assess the safety of compounds or drugs within living cells.
Other applications of iPSCs include their use as research products, as well as their integration into 3D bioprinting, tissue engineering, clean meat production, and potentially, wildlife conservation. Market competitors provide iPSC specific tools to scientists worldwide, including human iPSC lines and differentiated cells types, as well as optimized reagents, protocols, differentiation kits and more.
Technology allowing for the mass-production and differentiation of iPSCs in industrial-scale bioreactors is also advancing at breakneck speed. Finally, iPSC repositories provide researchers with the opportunity to investigate a diverse range of conditions using iPSC-derived cell types produced from both healthy and diseased donors.
Future Applications for iPSCs
Clearly, iPSC-derived cells represent a promising new technology platform. As their manufacture, administration, and safety profile improve, these cells will unquestionably usher in a new era of medicine.




















Tell Us What You Think!