Hemostemix Inc. (“Hemostemix”) (TSX Venture:HEM; OTCQB:HMTXF) is a publicly traded clinical-stage biotech company that develops and commercializes blood-derived stem cell therapies for medical conditions lacking adequate treatment options. Importantly, it is one of the first clinical-stage biotech companies to test a stem cell therapy within an international, multi-center Phase II clinical trial for patients with critical limb ischemia (“CLI”). CLI is a severe form of peripheral artery disease (“PAD”) that is caused by reduced blood flow to the extremities.
After showing promising results in a Phase I trial and with a large number of historical patient treatments performed in open trials, Hemostemix’s Phase II trial targets a participant’s diseased tissue with proprietary cells grown from his or her blood that can support the formation of new blood vessels. This lead cell therapy product is called ACP-01.
In early 2018, Hemostemix signed a contract with Aspire Health Science LLC (“Aspire”) for production of its lead cell therapy product, ACP-01, at Aspire’s FDA, certified good manufacturing practice (“cGMP”) compliant facility in Orlando, Florida. Building towards commercialization, Hemostemix has licensed the use, sale and import of ACP-01 for certain indications to Aspire, and it is continuing R&D activities for ACP-01 across cardiovascular, neurological and vascular indications.
Two key aspects differentiate Hemostemix within the cell therapy marketplace and make this a potentially disruptive technology. Firstly, it has world-class proficiency in peripheral blood stem cells, and these cells are sourced through a simple blood draw from the arm. Secondly, it controls a proprietary technology platform that can be applied across a wide range of disease indications.
Another strength is the company’s intellectual property assets. Hemostemix has secured a global portfolio of 50+ issued or pending patents that pertain to the collection, processing and utilization of peripheral blood stem cells. With clinical applications for peripheral blood stem cell still in its early stages, Hemostemix has secured this IP at an opportune time in history.
Because Hemostemix is pursuing a business opportunity with the potential to positively impact human health and medicine, I am excited to release this interview with Kyle Makofka, President and CEO. In this interview, we discuss the formation of Hemostemix, its product pipeline, clinical trial activity, and future directions. Mr. Makofka also explains Hemostemix’s peripheral blood platform technology and its clinical data demonstrating the ability of its autologous (self-derived) cell product to regenerate damaged tissue across a range of disease conditions.
Enjoy these insights from Mr. Makofka into how Hemostemix is developing personalized regenerative therapies with the potential to dramatically impact human health.
Interview with Kyle Makofka, President and CEO of Hemostemix Inc.
Cade Hildreth: What’s your professional background and how did you become involved with Hemostemix?
Kyle Makofka: My background has always been working with teams to drive operational efficiencies through internal management systems, mostly with companies that have a specialized technology or a unique business model. This can range from complete corporate restructuring or simply project management. I first became involved with Hemostemix when a longtime friend and business partner came to me with some concerns around an investment his Family Office had made in the company. He has a long track record of success and felt this organization had a disruptive technology but was underperforming in its execution. I was also intrigued by Hemostemix and its potential, as it did have what appeared to be an industry changing technology, however the company had not streamlined internal processes, lacked strategic vision and needed cash to support its growth trajectory.
When we first started to evaluate the company, I admit that I was very pessimistic. The thought of a cell-based technology with such a simple process that regenerated diseased and damaged tissue without medication or surgical intervention seemed unrealistic. With that in our mind we did a very deep dive into the struggling company. We traveled to the manufacturing lab in Israel, visited hospitals in Thailand where treatments were performed, spoke with company founders, scientists, cardiothoracic surgeons, interventional cardiologists and of course patients who had been treated with the company’s product. We tried to speak with as many people as we could that touched the technology, both within the company as well as third parties to get a fully comprehensive understanding.
What we uncovered was twelve years of history and development that led to a deep IP portfolio and a larger than expected library of patient data. The safety and efficacy appeared to be there and Hemostemix had documentation of over 300 patients being treated in open trials with congestive heart failure, angina pectoris, acute myocardial infarction and critical limb ischemia. As I do not have a medical background, the turning point for myself was the patient and doctor testimonials that demonstrated to me the technology had a positive impact and outcome for “no option” patients. I knew I wanted to be involved with this company as it was clearly a disruptive technology with significant potential to change how many different diseases could be treated using stem cell therapies. We came to the conclusion that the future lies in regenerative medicine, and Hemostemix was a compelling story with a significant head start. It was a company worth investing our time and resources into.
I joined the company as its Chief Restructuring Officer in January 2017, and with a lead order from a prominent businessman and his Family Office went out and raised much needed capital for the company to move forward. By September 2017 we had completely restructured the management team, resolved all outstanding issues and raised about $11 million. With a debt-free company, the following twelve months focused on getting the right team in place with scientific, corporate and public company expertise and defining the company’s short and long term strategies. This included specific research and development goals, streamlining operations and defining a path to commercialization. All of this led to getting our Phase II clinical trial for critical limb ischemia (“CLI’”) back up and running with Health Canada approval. We also succeeded in getting the trial approved with the US FDA which was extremely important. In addition, we simplified our manufacturing logistics by moving product manufacturing from the original lab in Israel to Orlando, Florida by partnering with Aspire Health Science. Aspire would be responsible for fixed cost manufacturing and continue research and development efforts. We checked a lot of boxes in a short period of time and it has been an incredible learning experience in an exciting industry, and that’s what has brought us to this point today.
Cade Hildreth: Wow, that is quite a story. When was Hemostemix formed and by whom?
Kyle Makofka: It was originally founded as a private company in 2006 in Toronto Canada, by Dr. Valentin Fulga. In 2014, Hemostemix went public on the TSX Venture Exchange.
Cade Hildreth: Excellent. Can you describe for me Hemostemix’s platform technology? For Hemostemix’s cell therapy product ACP-01, could you clarify for our readers what the acronym ACP stand for?
Kyle Makofka: Hemostemix’s platform technology is focused on an autologous (patient’s own) synergetic cell population (“SCP”). SCPs are basically a cell population within the blood consisting of adult stem cells and other supporting cell types. This cell population has the potential to generate various types of cellular products, depending on the culture conditions in which the cells are grown. This is a very broad platform from which various cell types can be generated from a very simple blood draw. Based on the synergetic cell population, we can differentiate these cells into a population of angiogenic cell precursors (“ACPs”) which induces blood vessel formation.
Angiogenic is term that means the formation of new blood vessels. Our lead product is ACP-01 for use in treatment of indications with diseased or damaged tissue that require some level of revascularization for patient recovery, including indications driven by peripheral arterial disease like critical limb ischemia. As you know, diabetes is becoming a worldwide epidemic and is one of the leading causes of PAD. Within the intellectual property platform we can also use this SCP to differentiate and expand into neural cellular precursors (“NCPs”) which could induce neural lineage cells.
We are currently focusing on commercialization of our treatment therapy for CLI now with ACP-01 within our current trial, but are also preparing a pre-IND submission for the use of ACP-01 in a heart trial, such as angina pectoris. Our next product initiative will be the development of NCP-01, our neural cellular precursor (NCP) product. To be specific, we are differentiating the SCP into that specific lineage.
As I mentioned earlier, with our technology platform, we are able to generate various types of cellular products, and depending on the culture conditions, the cells can be grown into various cell types, such as blood vessel cells, heart muscle-like cells and neural cells. This is what I feel differentiates us as a company that can offer individual custom cell therapies for potential multiple conditions. It is not a “blanket” cell therapy where you take something, say umbilical cord blood, and you pull out the mesenchymal stem cells (MSCs) and do an injection of that undifferentiated cell population.
We physically differentiate and expand the synergetic cell population. The angiogenic process lends itself to regenerating damaged tissue through a micro-vascularization process with new blood vessel formation. Additionally, we use flow cytometry and very precise quality control to know the exact makeup of the cell population that a patient gets re-injected with. This re-injection helps the body generate a micro-vascularization event. This is why we have seen success in both the heart and CLI. The angiogenic cell population is predetermined to induce healing in these types of ischemic conditions.
Cade Hildreth: Hemostemix’s technology platform potentially allows for the treatment of many disease indications. I know ACP-01 that is being explored in Phase II trials, and I believe that other clinical candidates, such as NCP-01 and BCP-01, are also under development by Hemostemix. Is this correct?
Kyle Makofka: Yes, this is correct. Hemostemix is now conducting a randomized, double-blind, placebo-controlled Phase II clinical trial for patients with CLI at sites within Canada and United States using our lead product ACP-01.
As mentioned earlier, when I came into the company, I needed to get up to speed rapidly, so I spoke as many people as I could who had a part in either developing or using this technology to get a full and unbiased perspective from every angle – from insiders to patients to doctors that had witnessed the treatment results. I was able to track down two of the original scientists who worked to patent the technology and learned from speaking with them that they had originally started with the neural cell product named NCP-01. For NCP-01, we would take peripheral blood from a blood draw, isolate the SCP, and then use our intellectual property and related protocol to differentiate the cells into neural cell precursors. This NCP-01 R&D even progressed to completion of a small animal study. Unfortunately, the early pioneers of our technology found that they had a very long and expensive road ahead.
That was back in the early 2000s when cell therapy was still in its infancy. This NCP-01 program was therefore put on hold and they focused their efforts on angiogenic precursor cells, used in ACP-01, as this had a shorter manufacturing time, and also proliferated and differentiated very well. This became the lead product and the start of what is today Hemostemix.
Now that we have ACP-01 into Phase II trials, we have gone back and are continuing R&D on our NCP-01 product, because if it is successful, that puts the company in an entirely different category compared to other stem cell companies. We could then potentially treat diseases like Parkinson’s, Alzheimer’s, ALS, Dementia, Multiple Sclerosis and possibly many other neurological conditions.
If our success with NCP-01 is anything like the success we are seeing with ACP-01, I cannot begin to tell you what that would mean for the company and for medicine.
Cade Hildreth: Why did you choose to target Critical Limb Ischemia (CLI) for Hemostemix’s Phase II clinical trial of ACP-01?
Kyle Makofka: One of the pioneers of the technology, Dr. Fulga, recognized that CLI was an orphan disease that has limited treatment options that could be a much faster pathway to commercialization of ACP-01. Once you have exhausted standard of care and surgical revascularization options for CLI, the next treatment option for that patient is amputation. After amputation occurs, medical data shows that morbidity rates go up drastically for this patient population.
The majority of our historical patient data from international treatments was actually for the heart, as ACP-01 was used to treat over 300 cardiac patients in open trials. Like cardiac conditions, CLI is also related to blood flow restrictions that causes damaged tissue, so we felt that ACP-01 could also be used to treat CLI. Should we see similar safety and efficacy data from our trial as we saw in those cardiac patients, we could have the only alternate treatment for a patient population facing life-changing amputation. In the cardiac disease space there are multiple devices and multiple drug trials competing for the same patient population. With CLI, there are limited treatment options and very few companies focusing on CLI trials utilizing stem cells. The CLI market is surprisingly large and growing, with many of these patients having absolutely no option other than amputation. To provide a viable treatment where there is none, is a platform to build a business.
Cade Hildreth: Wow, that was a powerful decision. I’m curious, in your opinion, what differentiates Hemostemix from other stem cell companies?
Kyle Makofka: Our main differentiators are the source of our cells and the robust nature of the IP platform we have developed. We have the ability to scale up with different cell types to treat multiple indications from a single platform. We have publications and open trial data on over 300 patients treating multiple indications with a level of safety and efficacy. This is done from a single technology platform that uses peripheral blood as its cell source.
I’m sure you’ve seen through your work with BioInformant that bone marrow and adipose or fat tissue have received a lot of attention from the cell therapy sector, as well as some of the birth tissues like umbilical cord blood and tissue. Unfortunately, bone marrow collection is an invasive and painful procedure, adipose tissue requires liposuction by an experienced physician, and birth tissues require long-term cryopreservation followed by thawing of the cells which can damage the cell population. My point is, all of these approaches have significant limitations that you do not find with peripheral blood.
In contrast, Hemostemix’s advantage is that people are familiar with getting blood drawn from their arm, because it is a very common procedure. It is safe and effective, non-surgical, has low patient risk, avoids ethical concerns, and is available through self-donation for the autologous, or patient’s own blood, protocol.
From the business perspective, scalability and manufacturing have proven to be major roadblocks for many cell therapy companies. In most cases it becomes very expensive to scale due to the specialized people and equipment required to do this.
In contrast, with Hemostemix, a blood draw from the arm is processed in our laboratory using our proprietary manufacturing protocol and is then returned back to the body. This manufacturing protocol is a simple overall process which lends itself to optimization and automation in the near future. As part of our ongoing R&D work, we are working on a strategy that will allow us to get to a point where we can automate our entire process and have it available at key medical locations for on-site manufacturing.
Currently, manufacturing time for our ACP-01 cell therapy takes five days, and with medical courier times to our laboratory and back, current treatment occurs 7 days from the date blood is drawn. However, our internal research has recently proven that manufacturing time can be reduced to only three days, which we plan to get regulatory approval for in order to utilize this optimized process in our current clinical trial. What that translates to is a process that can be automated. This is a requirement for worldwide commercialization.
Imagine that anybody could go in and get their blood drawn at a doctor’s office and then get treated shortly thereafter at the same site. Our ultimate goal would be to have blood collection, manufacturing, and patient treatment all occur in one spot. That is something that really excites us.
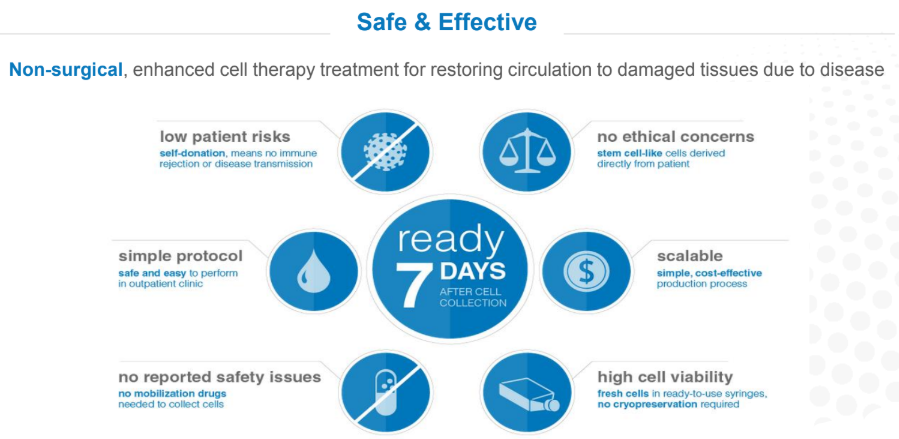
Cade Hildreth: To summarize the key points, this blood draw is a simple and straight forward process, there is a very short manufacturing process that could be as little as three days, there is the potential for automated manufacturing, and ultimately, the result is a personalized regenerative cell therapy.
Kyle Makofka: Exactly.
Cade Hildreth: What are the differences associated with the manufacturing of your neural cellular precursors (NCP-01) versus angiogenic cellular precursors (ACP-01)?
Kyle Makofka: NCP-01 has a more difficult differentiation and expansion process, and these cells take substantially longer to develop. At this time, our neural cellular precursors need over six weeks of incubation and culturing to get them to differentiate into neural lineage cells.
Again, it is still based on that same technology platform used for ACP-01, but with NCP-01, we have to re-create the environment of the body and mimic natural processes to guide cellular differentiation.
Cade Hildreth: What do you anticipate will be Hemostemix’s timeline for the commercialization of ACP-01, which is currently in Phase II trials for CLI?
Kyle Makofka: I am very excited about our pathway to commercialization for ACP-01, which we have several strategies for. Our Phase II trial for CLI is currently well underway in the U.S. and Canada, which we estimate should be completed in 2019. Then we intend to move immediately into Phase III trials.
There is the potential for regulatory bodies to view CLI as an orphan disease (one with limited treatment options) and decide to accelerate the trial process based on our results. If that’s the case, we could get into a Phase III trial earlier than expected.
We have also entered into a licensing agreement with our contract manufacturing partner Aspire Health Science, which gives Aspire exclusive rights to use and sell ACP-01 in the Bahamas, Costa Rica, the Dominican Republic, Mexico, Panama, and the State of Florida for use in approved open trials and compassionate care cases. Some of these indications include critical limb ischemia, coronary artery disease, peripheral artery disease, and congestive heart failure.
Hemostemix is committed to focusing on commercializing ACP-01 through formal regulatory pathways within the United States and Canada. However, we have such a large body of safety and efficacy data on our product, we are comfortable writing an expanded access policy, which would allow doctors with patients that have critical life-threatening diseases with no other treatment options to be able to access our stem cell therapy.
Aspire has worked with the Bahamas Ministry of Health and Dr. Conville Brown in Nassau, Bahamas, to start an open trial for congestive heart failure (CHF) involving 20 patients and an open trial for 20 patients for CLI. It is important to mention that all of the data collected by Aspire belongs to Hemostemix.
Also, if there is a physician working on a compassionate care case who feels they have a patient that fits the expanded access policy as we discussed earlier, they could potentially treat that patient outside of the current trial through a compassionate care application, or that patient can qualify for open trial study through Aspire, our licensing partner.
Cade Hildreth: From a business perspective, what is the importance of Hemostemix’s platform technology and its ability to be leveraged across a wide range of disease indications?
Kyle Makofka: This is an extremely important question. Once Hemostemix’s ACP-01 product gets validated during Phase II and III trials, we could potentially expand our peripheral blood technology platform into almost any cell lineage, which is incredibly exciting. For example, I have already spoken about ACP-01 for heart indications and NCP, but we could also pursue osteogenic (bone) precursors, which we have not yet invested time or resources into.
When you look at the other stem cell companies, they have a specific formulation for a specific indication. If the indication changes, then they must go back to square one with a new formulation and undertake animal safety testing and spend another 10-15 years and hundreds of millions of dollars to fully develop.
I cannot emphasize this message clearly enough. ACP-01 is a platform that has shown safety and efficacy in treating multiple indications without severe adverse events. ACP-01 utilizes the same process for the heart that we are using to treat CLI in the leg. Nothing changes except the indication, so we are not going back to square one every time and incurring costly development time or money. Unlike other technologies, our ACP-01 technology platform has the potential to be widely commercialized across many indications. It is that robust.
Cade Hildreth: The “one platform, many disease” angle is a powerful differentiator for Hemostemix. What are your 3-5 year goals for the company?
Kyle Makofka: The short-term goal of Hemostemix is to also move into a Phase II, and maybe even a Phase III trial, for a cardiac disease indication such as angina pectoris. We are currently working on a pre-IND submission on this for the FDA which we expect to complete shortly. Because we have collected so much safety data on ACP-01 over the years, we are able to explore and pursue other indications. This could also include idiopathic pulmonary hypertension, because it is an orphan disease and patients have no other treatment options.
Long-term, our goal is to commercialize our neural cell precursor technology as well, and determine the best suited indication for it, because if it has anywhere near the level of success that we have witnessed with ACP-01, then the medical world has to have it.
Within the next few years, I anticipate that we will have commercialized ACP-01 for at least two indications and have NCP-01 into human clinical trials.
Cade Hildreth: One of the things that caught my attention when I was digging into Hemostemix as a company was the depth and the breath of the patent portfolio. Could you tell me more about intellectual property (IP) that the company has secured?
Kyle Makofka: Yes. Again, you know we’ve primarily focused on ACP-01 to date, but Hemostemix did a fantastic job early in the company’s history of identifying all aspects of our manufacturing process and protecting it. Therefore, our IP spans five different patent families with over 50 patents issued and pending worldwide that cover the regulation of the cells themselves, as well as the manufacture and differentiation of these cells.
Also, Hemostemix formed patent groups and took them worldwide. We have a European Union (EU) patent group, a U.S. patent group, Mexico and Canadian patent group, so we are very well protected and still has quite a nice lifespan left on our IP. We can build from that now, because all of those patents are in place. We feel our ongoing R&D work will also result in further additions to our IP portfolio.
Cade Hildreth: What role will Hemostemix’s manufacturing process play in the company’s long-term success?
Kyle Makofka: For many biotech companies, their cost of manufacturing gets so high that it becomes very difficult to get the cell therapy product commercialized.
In my opinion, two things must happen for a cell therapy company to be successful:
- The manufacturing protocols design and processing times must be optimized and shortened.
- The platform needs to be moved to an automated manufacturing process that can be put into medical centers.
If we can position ourselves with automated manufacturing processes that can be introduced into medical centers worldwide, this would be significant for successful commercialization. In contrast, many cell therapy companies struggle with a difficult collection process, weeks of processing, difficult logistics, cryopreservation, and the need for medical couriers. These companies are restricted when it comes to treating hundreds or thousands of patients in a realistic time line.
With Hemostemix, we are already looking at how to treat millions of patients, because that’s how many are out there with medical needs, especially as we witness the rise in peripheral arterial disease and diabetes, which is out of control, not only in North America but also worldwide. We have to be able to scale as quickly as the problem, and right now, the problem is dramatically out-scaling us.

Cade Hildreth: I understand you currently use an autologous process for ACP-01. Is it correct that your recent R&D has shown your technology works on an allogeneic process as well?
Kyle Makofka: Yes, currently we use an autologous process, meaning that the ACP-01 cell therapy is produced from a patient’s own blood. We have been utilizing Aspire’s lab and personnel to run 39 research batches over the past several months to further our technology platform. We made some exciting progress not only for our manufacturing optimization, but also in identifying that we can further expand our ACP-01 technology process to also support an allogeneic process. In short, an allogeneic process involves the use of donor or third-party blood that can be used processed and then used to treat a patient. Some patients may be too ill or suffer comorbidities (having multiple diseases) which may result in a poor blood sample for processing and ultimately may not be treatable.
Being able to use healthy donor blood would allow for the potential ability to treat all patients, including those who are very ill where an autologous treatment is not optimum, which would definitely expand our commercialization potential. We plan to obtain approval from the US FDA and Health Canada to also use this allogeneic process by doing a safety trial or adding an allogeneic safety arm to our current Phase II clinical trial for CLI. We feel having both an autologous and allogeneic process will position us well as a leader in this field. If you look at several other stem cell companies with high market valuations, many use an allogeneic process. Now we have both, which further adds to the flexibility and breadth of our technology platform.
Cade Hildreth: That is a very exciting achievement. On a personal basis, what excites you most about Hemostemix?
Kyle Makofka: I strongly believe it is an impact investment, meaning we can build a successful business by developing technology with the potential to positively impact millions of people who suffer from debilitating disease. Hemostemix is a rare and exciting opportunity. As you know from following the cell therapy industry over the past decade, it is extremely rare to see an effective platform for multiple indications that is also a platform that has the potential to be commercialized and scaled. Because it is based on a simple blood draw, our technology has the capacity to become a new standard of medical care across a diverse range of indications and combined therapies.
Cade Hildreth: What is the market capitalization of Hemostemix?
Kyle Makofka: We are at approximately USD$27 million. (TSX VENTURE: HEM; OTCQB: HMTXF). Prior to the restructuring and financing we had a market cap low of approximately $4-5 million. Although we have seen our market cap steadily increase over the past 12 months, I feel we are still incredibly undervalued for what we have. To add context, other stem cell companies with products in Phase II clinical trial have market caps of $250 million and up.
Cade Hildreth: That represents incredible upside potential for Hemostemix. Alright, the most important question of all. How can people learn more about Hemostemix?
Kyle Makofka: Your audience can reach out to me or anyone else on the Hemostemix team through our website, www.Hemostemix.com, or they can email us at [email protected].
Cade Hildreth: Fantastic. Thank you so much for connecting and for sharing these valuable insights.
Kyle Makofka: You do a fantastic job getting the message out there and helping to educate people about regenerative medicine and stem cell therapies. I truly appreciate what you are doing and the importance of it to the future of medicine. I thank you as well.
Do you have questions about Hemostemix, including its platform technology or opportunities for commercialization? Ask them in comments below.
Disclaimer:
This article is for informational purposes only. The information contained in this article is subject to change without notice, and Hemostemix Inc and the author assume no responsibility to update the information contained in this article. The information contained within this article should not be construed as offering of investment advice. Those seeking direct investment advice, should consult a qualified, registered, investment professional. This is not a direct or implied solicitation to buy or sell securities. Readers are advised to conduct their own due diligence prior to considering buying or selling any securities. The information in these article is historical in nature, has not been updated, and is current only to the date indicated in the particular article. This information may no longer be accurate and therefore you should not rely on the information contained in this article. To the extent permitted by law, Hemostemix Inc. and its employees, agents and consultants exclude all liability for any loss or damage arising from the use of, or reliance on, any such information, whether or not caused by any negligent act or omission. BioInformant.com is not engaged in an investor relations agreement with Hemostemix Inc., however, it did receive compensation from Hemostemix Inc. for the preparation and distribution of this article. Cade Hildreth had no stock position in Hemostemix Inc. at the time of publication. BioInformant also has no stock position in the company noted in this article.




















Outstanding technology designed to safely and simply stimulate regeneration of healthy body tissue in this case blood vessels
Great that it has already been tested on 300 patients or more and is effective and safe.
Also world renowned physicians are on the advisory board.
I found this article very concise and informative and engaging
Also the concept of an on sight lab in major hospitals to offer regenerative medicine is GROUND BREAKING