Key Points:
- Natural killer (NK) cells represent innate immune cells with potent antitumor activity.
- Glycostem Therapeutics has established an innovative production process that yields high numbers of allogeneic NK cells with potent activity towards both hematologic and solid malignancies which can be used “off-the-shelf”.
- In a first-in-human phase I study in elderly patients with acute myeloid leukemia, these NK cells were well tolerated, had a favorable toxicity profile and were associated with promising efficacy.
- The possibility to genetically engineer the NK cells with chimeric antigen receptors or T cell receptors offers additional opportunities in immuno-oncology.
Immunotherapy
In recent years, immunotherapy has quickly developed into the fourth treatment modality in cancer, complementing surgery, chemotherapy and radiotherapy. Indeed, due to long-term efficacy and favorable toxicity profiles various immunotherapies have become a standard treatment in an ever growing number of solid and hematologic malignancies.
When classifying the current immunotherapies applied in cancer patients, two major groups can be distinguished: active immunotherapies and adoptive immunotherapies. By inducing antitumor immune responses in the host, active immunotherapies, such as immune checkpoint inhibitors and vaccines, contribute indirectly to cancer eradication.
In contrast, adoptive immunotherapies, including the adoptive transfer of NK cells, T cells and cytotoxic antibodies, can directly kill cancer cells.
NK cells
NK cells are innate lymphoid cells with potent antitumor activity.1 Although they share a number of developmental, phenotypical and functional features with (cytotoxic) T cells, NK cells have multiple unique characteristics as well. For instance, they can be readily identified in the peripheral blood and (lymphoid) tissues through the expression of CD56 and CD335, and lack of CD3 and T cell receptors.
Importantly, the cytotoxic activity of NK cells does not depend on the recognition of a specific antigen presented by a major histocompatibility complex (MHC), a typical prerequisite for T cells. Instead, the activation of NK cells is tightly regulated by a substantial number of stimulatory and inhibitory receptors, including the natural cytotoxicity receptors and killer cell immunoglobulin-like receptors (KIRs).2
Thus, the joint signaling of these receptors determines if an NK cell becomes activated or not. Since tumor cells and virally infected cells frequently display an altered phenotype, such as diminished or absent expression of MHC, NK cells generally become activated by these aberrant cells.
This results in eradication of the aberrant cells and in the release of proinflammatory cytokines, chemokines and growth factors that support the responses of other immune cells. At the same time, the reactivity of NK cells towards healthy tissues is limited, as exemplified by their favorable toxicity profile in clinical studies.
NK cells for cancer immunotherapy
In view of their potent antitumor activity, NK cells have been extensively studied in clinical studies involving patients with hematologic and solid cancers. Where initial clinical studies with autologous NK cells yielded disappointing results, the action of allogeneic NK cells proved to be very beneficial in patients with hematologic malignancies.
In 2002, a study by Ruggeri et al. indicated for the first time that, following allogeneic hematopoietic stem cell transplantation (alloHSCT), KIR-dependent alloreactivity of NK cells was associated with decreased leukemia relapse and graft rejection in patients with acute leukemia.3
Subsequent studies demonstrated that adoptive transfer of allogeneic NK cells in patients with hematologic cancers is safe and efficacious. For instance, a landmark study by Miller et al. showed that infusion of allogeneic, haploidentical NK cells plus interleukin 2 was safe and associated with anti-leukemic activity in a subset of poor-prognosis patients.4
Lymphodepletion with cyclophosphamide and fludarabine prohibited rejection of the allogeneic NK cells and allowed their in vivo persistence and expansion, highlighting the importance of immunosuppression in the non-transplant setting. In the following years, a substantial number of clinical studies demonstrated that in transplanted patients the adoptive transfer of haploidentical NK cells is well tolerated and safe.5
Moreover, treatment was associated with alloreactive responses towards various hematologic malignancies, including acute myeloid leukemia (AML), myelodysplastic syndrome and non-Hodgkin lymphoma. These encouraging results propelled the evaluation of NK cell infusion in patients with solid tumors.5
Where initial studies in renal, breast and ovarian cancer were generally disappointing, a combination of NK cell transfer, targeted therapy and chemotherapy was associated with clinically meaningful responses in children with neuroblastoma.4,6,7 Successive studies indicated that the outcomes in patients with solid tumors might be improved by cytokines, KIR-blocking antibodies and immune checkpoint inhibitors.8
Another promising option to increase the efficacy of NK cells is to genetically modify these cells with chimeric antigen receptors (CARs), or bispecific NK cell engagers, such as those simultaneously binding CD16 on NK cells and EpCAM on carcinomas.
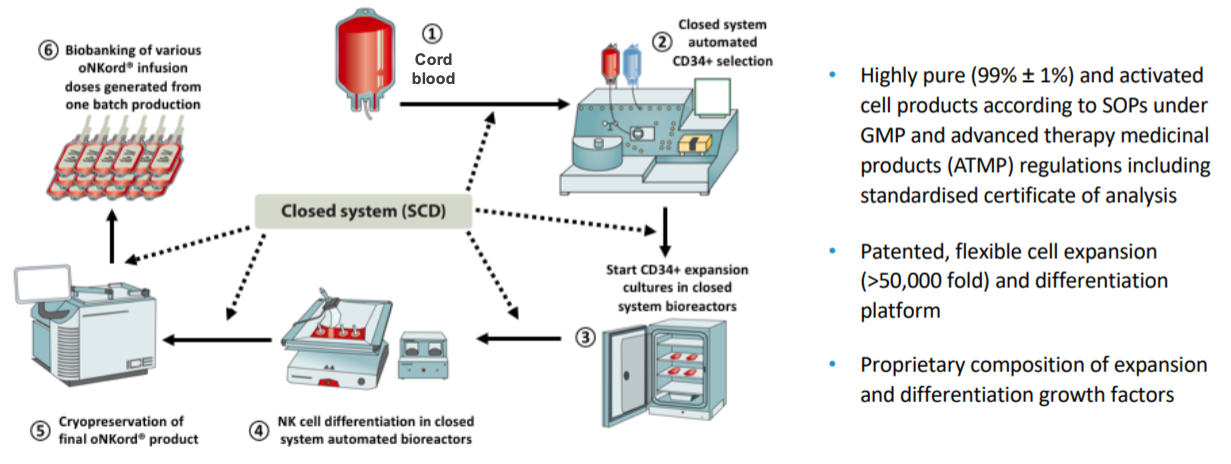
Glycostem’s platform for the generation of large numbers of cancer reactive NK cells
For the clinical studies described above, primary NK cells were generally selected from leukapheresis products. Unfortunately, this selection process has several disadvantages, including a frequently limited and/or inconsistent yield of NK cells with varying purity and activation status.
To circumvent these disadvantages, Glycostem has successfully developed an innovative platform to expand and differentiate clinical-grade NK cells from hematopoietic stem and progenitor cells (HSPCs) isolated from umbilical cord blood (UCB; Figure 1).
In addition, Glycostem developed the unique and clinical grade culture medium ‘GBGM’ which optimizes the culture process. The generation of NK cells from UCB-HSCs has the advantage that UCBs are readily available through UCB banks. In addition, Glycostem’s patented UCB-NK cell platform has proven to consistently yield large numbers clinical-grade NK cells that resemble NK cells from peripheral blood.9-11
The >3-log expansion of the cells allows the infusion of a greater number of NK cells and/or permits multiple infusions. Functional in vitro studies demonstrated that this NK cell product, named ‘oNKord®’ efficiently kills various tumor cell lines and primary leukemia cells at low NK-target ratios. In mouse models, the NK cells of oNKord® migrated to various (lymphoid) organs, including the bone marrow, spleen and liver, upon infusion.12
Furthermore, the simultaneous administration of low-dose IL-15 mediated NK cell persistence and expansion. Importantly, a single infusion of oNKord® efficiently targeted human leukemia cells, resulting in significantly prolonged survival. These positive results warranted the evaluation of oNKord® in clinical studies involving patients with cancer.
For the treatment of patients with AML, oNKord® received an Orphan Drug Designation from the Committee for Orphan Medicinal Products of the European Medicines Agency in 2014 and the Food and Drug Administration (FDA) in 2016.
Infusion of oNKord® is feasible, safe and efficacious in patients with leukemia
Previously, a first-in-human phase I dose-escalation study determined the feasibility, safety and efficacy of oNKord® in elderly patients with AML who were not eligible for alloHSCT.
Hereto, ten patients in morphologic complete remission after first or second line chemotherapy or hypomethylating agents received a single infusion of escalating doses of oNKord® (between 3 and 30 x 106 NK cells/kg body weight). To improve NK cell persistence, patients were conditioned with non-myeloablative cyclophosphamide and fludarabine from day -6 to -3. Safety was the primary endpoint.
The results showed that oNKord® was well tolerated and had a favorable toxicity profile.13 Importantly, treatment with oNKord® did not lead to graft-versus-host disease (GvHD), a major cause of morbidity and mortality in patients treated with allogeneic T cells. Despite the fact that no cytokine boosting was given to the patients, the NK cells were readily detected in the peripheral blood between infusion and day 8, which was accompanied by augmented IL-15 levels in the plasma.
In addition, upon infusion of oNKord® a donor chimerism of up to 3.5% was found in the bone marrow. Phenotyping of the injected NK cells showed that they had further matured in vivo, as indicated by the acquisition of CD16 and KIR expression.
Importantly, two of four patients with minimal residual disease (MRD) in their bone marrow at baseline became MRD negative following administration of oNKord®. Additional efficacy analyses suggested that the overall survival in the total cohort as compared to a historical control cohort was strongly improved (Figure 2).
These results demonstrated that the adoptive transfer of oNKord® in AML patients is feasible, well tolerated, safe and associated with substantial and clinically relevant efficacy. Furthermore, they warrant future studies investigating the application of oNKord® as adjuvant, consolidation or combination therapy or as a bridge to transplant in AML patients.
Glycostem is conducting a phase I/IIa multicentre trial in Europe for the evaluation of the safety and efficacy of oNKord® repeat infusions – three infusions four days apart – in patients with AML who are in complete morphologic remission with MRD, and who do not have a strong indication for alloHSCT. The evaluation of MRD as a surrogate endpoint for overall survival is key to demonstrate the efficacy of oNKord® as consolidation therapy. Further clinical development in AML is planned in the United States of America (USA).
In addition, when preliminary data about oNKord® repeat infusions in patients with AML are available, Glycostem will conduct a phase IIa multicentre trial in Europe for the evaluation of the safety and efficacy of oNKord® repeat infusion in patients with refractory multiple myeloma (MM).
On the basis of the outcomes of the phase I/IIa trials in MM and AML, Glycostem plans to conduct confirmatory comparative phase III trials, and to file Investigational New Drug applications to the FDA.
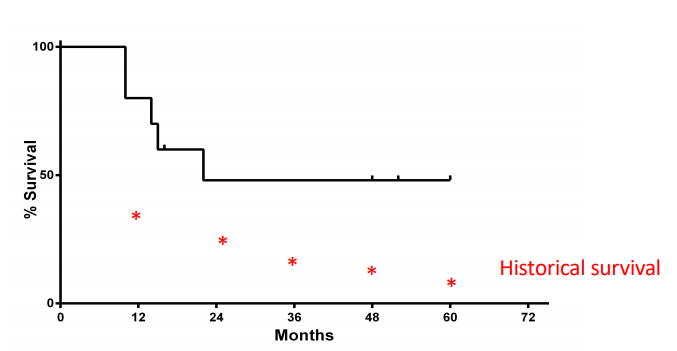
*Data on the survival of historical controls, patients of 65-74 years old diagnosed with AML between 2001 and 2010, were derived from the Netherlands Cancer Registry.
oNKord® shows activity against solid tumors
In addition to hematologic cancers, oNKord® has shown significant activity against solid tumors. For instance, oNKord® displayed significant cytotoxic efficacy against colon cancer cells with or without aberrations of EGFR, RAS and BRAF.14 This efficacy was confirmed in a preclinical mouse model, showing that oNKord® significantly prolonged the survival of mice with colon cancer.
In another study, oNKord® effectively killed cervical cancer cells, which was dependent on the expression of the NK cell-activating receptors NKG2D and DNAM-1, but independent of HLA class I expression.15 Of note, oNKord® compared to allogeneic NK cells derived from peripheral blood demonstrated a superior cytotoxic activity against both colon cancer and cervical cancer cells.14,15
These positive results in preclinical studies paved the way for Glycostem’s clinical studies evaluating the safety and efficacy of oNKord® in patients with solid tumors (Figure 3). A single center, phase I, dose ranging clinical trial investigating an alternative for the cyclophosphamide and fludarabine conditioning regimen for oNKord® infusion is planned in patients with various advanced solid tumors.
This trial will be followed by a phase I/II study of oNKord® in patents with selected solid tumors.
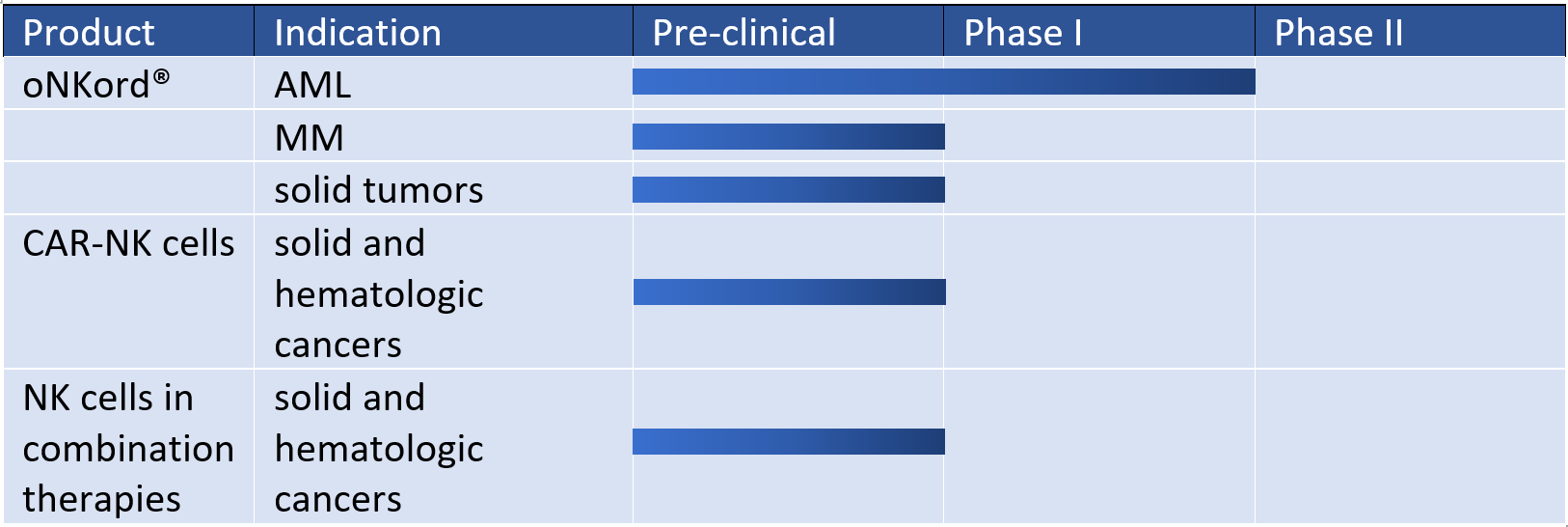
Glycostem’s complementary off-the-shelf NK cell products in immuno-oncology
The establishment of oNKord® as a safe and efficacious cellular immunotherapy opens possibilities for the development of combination therapies involving off-the-shelf NK cell products for immuno-oncology. For instance, combinations with other treatment modalities, such as targeted therapies and immune checkpoint blockade, are appealing and being explored by Glycostem.
In addition, by means of genetic engineering, Glycostem is currently developing NK cells expressing CARs for various molecular targets, such as OGD2 in glioblastoma and HSP70 in various solid tumors. There is increasing evidence that these CAR-NK cells hold clinically relevant activity against hematologic and solid malignancies while sparing normal tissues.16
For instance, a recent phase I/II study showed that infusion of CD19-specific CAR-NK cells was associated with a 73% response rate in patients with relapsed or refractory CD19-positive lymphoid cancers.17 Glycostem, in collaboration with others, demonstrated that transduction of NK cells with a CAR against CD123, as a surrogate cancer-associated antigen, significantly increased their cytotoxic capacity against CD123-expressing cancer cells.17
These encouraging results illustrate the complementary opportunities in immuno-oncology with NK cell products, such as oNKord®.
Conclusion
During the past years Glycostem has established itself as a leading company in the generation of innovative cellular immunotherapies for cancer. Cornerstone of these therapies is oNKord®, an off-the-shelf, sizeable cellular therapy product based on allogeneic NK cells. These tumor reactive immune cells can be generated in large numbers and high purity by Glycostem’s unique and efficient technology platform.
In a first-in-human dose escalation trial conducted for the evaluation of the safety and tolerability of oNKord® in patients with AML, some promising signals of efficacy were collected. Furthermore, genetic engineering of oNKord® opens possibilities for the generation of additional therapies, including those based on NK cells expressing chimeric antigen receptors.
Glycostem is involved in various partnerships with biotech and pharma companies for co-development or licensing out. Private placement and Initial Public Offering are planned by Glycostem from 2021 onwards.
References
1. Chiossone L, et al. Natural killer cells and other innate lymphoid cells in cancer. Nat Rev Immunol 2018;18:671-88.
2. Long E, et al. Controlling natural killer cell responses: integration of signals for activation and inhibition. Annu Rev Immunol 2013;31:227-58.
3. Ruggeri L, et al. Effectiveness of donor natural killer cell alloreactivity in mismatched hematopoietic transplants. Science 2002;295:2097-100.
4. Miller J, et al. Successful adoptive transfer and in vivo expansion of human haploidentical NK cells in patients with cancer. Blood 2005;105:3051-7.
5. Shimasaki N, et al. NK cells for cancer immunotherapy. Nat Rev Drug Discov 2020;19:200-218.
6. Geller MA, et al. A phase II study of allogeneic natural killer cell therapy to treat patients with recurrent ovarian and breast cancer. Cytotherapy 2011;13:98-107.
7. Federico SM, et al. A pilot trial of humanized anti-GD2 monoclonal antibody (hu14.18K322A) with chemotherapy and natural killer cells in children with recurrent/refractory neuroblastoma. Clin Cancer Res. 2017;23:6441-9.
8. Nayyar G, et al. Overcoming resistance to natural killer cell based immunotherapies for solid tumors. Front Oncol 2019;9:51.
9. Spanholtz J, et al. High log-scale expansion of functional human natural killer cells from umbilical cord blood CD34-positive cells for adoptive cancer immunotherapy. PLoS One 2010;5: e9221.
10. Spanholtz J, et al. Clinical-grade generation of active NK cells from cord blood hematopoietic progenitor cells for immunotherapy using a closed-system culture process. PLoS One 2011;6:e20740.
11. Lehmann D, et al. Ex vivo generated natural killer cells acquire typical natural killer receptors and display a cytotoxic gene expression profile similar to peripheral blood natural killer cells. Stem Cells Dev 2012;21:2926-38.
12. Cany J, et al. Natural killer cells generated from cord blood hematopoietic progenitor cells efficiently target bone marrow-residing human leukemia cells in NOD/SCID/IL2Rg(null) mice. PLoS One 2013;8:e64384.
13. Dolstra H, et al. Successful transfer of umbilical cord blood CD34+ hematopoietic stem and progenitor-derived NK cells in older acute myeloid leukemia patients. Clin Cancer Res 2017;23:4107-18.
14. Veluchamy JP, et al. In vivo efficacy of umbilical cord blood stem cell-derived NK cells in the treatment of metastatic colorectal cancer. Front Immunol 2017;8:87.
15. Veluchamy JP, et al. High-efficiency lysis of cervical cancer by allogeneic NK cells derived from umbilical cord progenitors is independent of HLA status. Cancer Immunol Immunother 2017;66:51-61.
16. Hodgins JJ, et al. Killers 2.0: NK cell therapies at the forefront of cancer control. J Clin Invest 2019;129:3499-3510.
17. Liu E, et al. Use of CAR-transduced natural killer cells in CD19-positive lymphoid tumors. N Engl J Med 2020;382:545-53.
18. Oberschmidt O, et al. Development of automated separation, expansion, and quality control protocols for clinical-scale manufacturing of primary human NK cells and alpharetroviral chimeric antigen receptor engineering. Hum Gene Ther Methods 2019;30:102-20.
Source: Glycostem


Glycostem could non-invasively deliver and target their NK cells to treat brain tumors, including glioblastoma, using the intranasal method we developed that bypasses the blood-brain barrier. We would be happy to collaborate and/or assist them in doing so.