 Cynata Therapeutics Ltd (ASX: CYP) is a clinical stage regenerative medicine company specializing in stem cell therapeutics that has a proprietary technology for manufacturing induced pluripotent stem cell (iPSC) derived mesenchymal stem cells (MSCs). Cynata is the only company in the world that can mass-produce therapeutic mesenchymal stem cells deriving from a single donor at a consistent quality and affordable cost.
Cynata Therapeutics Ltd (ASX: CYP) is a clinical stage regenerative medicine company specializing in stem cell therapeutics that has a proprietary technology for manufacturing induced pluripotent stem cell (iPSC) derived mesenchymal stem cells (MSCs). Cynata is the only company in the world that can mass-produce therapeutic mesenchymal stem cells deriving from a single donor at a consistent quality and affordable cost.
For those unfamiliar with Cynata, Fujifilm took a A$3.97 million strategic equity stake in the company in January 2017. Under the partnership, Cynata Therapeutics and Fujifilm agreed to collaborate on the further development and commercialisation of Cynata’s lead Cymerus™ therapeutic MSC product CYP-001 for graft-versus-host disease (GvHD). The deal also included potential future upfront and milestone payments in excess of A$60 million and double-digit royalties on Cynata’s CYP-001 product net sales.
There are several reasons why Fujifilm was attracted to Cynata Therapeutics. Namely, Cynata achieved two world firsts by:
- Developing technology to enable the mass-production of therapeutic grade stems cells
- Receiving approval to commence an iPSC-derived allogeneic MSC clinical trial
World’s First Trial of an Allogeneic iPSC-Derived Therapeutic Product
In September 2016, Cynata announced it received advice from the UK Medicines and Healthcare products Regulatory Agency (MHRA) that its Phase I clinical trial application had been approved. The announcement reverberated worldwide, because it allowed Cynata to initiate the world’s first clinical trial involving a therapeutic product derived from allogeneic (unrelated to the patient) induced pluripotent stem cells (iPSCs).
Titled “An Open-Label Phase 1 Study to Investigate the Safety and Efficacy of CYP-001 for the Treatment of Adults With Steroid-Resistant Acute Graft Versus Host Disease,” the trial was initiated in March 2017 (Clinical Trial ID: NCT02923375).
With sites in Australia and the UK, the trial is a multi-centre, open label, dose escalation study designed to assess the safety, tolerability and efficacy of two infusions of CYP-001, in adults who have steroid-resistant GvHD.
Cynata has also collected strong data in multiple pre-clinical studies for the treatment of asthma, heart attack, and critical limb ischemia (CLI), areas that it will likely pursue as forthcoming clinical targets.
CYP-001 Trial for GvHD
Cynata’s Phase I clinical trial pertains to the treatment of GvHD (Graft vs. Host Disease) in bone marrow transplant patients. Participants who enrolled are adults who underwent an allogeneic haematopoietic stem cell transplant (HSCT) to treat a haematological disorder and were subsequently diagnosed with steroid-resistant Grade II-IV GvHD.
The primary objective of the trial was to assess safety and tolerability, while the secondary objective was to evaluate the efficacy of two infusions of CYP-001 in adults with steroid-resistant GvHD.
Within the trial, efficacy was assessed on the basis of response to treatment (as determined by change in GvHD Grade) and overall survival at 28 and 100 days after the administration of the first dose.
Participants are also being followed for up to two years under a separate non-interventional study protocol.
Given the excitement about the far-reaching clinical potential of iPSC derived products, the trial is a major advance for the field. It establishes a sound basis for bringing iPSC derived products – not just MSCs – into the clinic. That is a salient achievement for researchers, clinicians, corporates and patients alike.
Completion of Primary Evaluation Period for Patients in Cynata’s Phase 1 Trial
On August 30, 2018, Cynata Therapeutics announced the completion of the Primary Evaluation Period for all 15 patients enrolled in its Phase 1 clinical trial of CYP-001, including patients from both Cohort A and Cohort B.
As highlighted in Cynata’s press announcement, CYP-001 met all safety and efficacy endpoints in the trial.
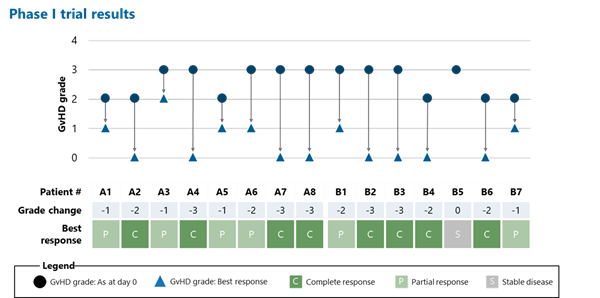
Most importantly for the patients in the trial, their families and caregivers, 14 out of 15 patients showed an improvement in GvHD severity by at least one grade compared to baseline. This is in marked contrast to the typically poor prognosis for such patients.
It is also noteworthy that GvHD signs and symptoms completely resolved in 8 out of 15 patients.
As stated by Dr Kilian Kelly, Cynata’s Vice President, Product Development:
“We are pleased to announce that CYP‑001 has met all clinical endpoints in the first trial of a product based on our Cymerus stem cell technology platform, which validates its potential to generate safe and effective iPSC-derived MSC therapies. Steroid-resistant GvHD is a devastating disease with a high rate of mortality. The clinical results from patients in Cohorts A and B are highly encouraging, as all of the patients had failed to respond to corticosteroid therapy, the only approved treatment for GvHD. We look forward to advancing our Cymerus MSCs into Phase 2 trials for GvHD and other indications.”
Significance of Cynata’s CYP-001 Safety and Efficacy Data
The significance of Cynata’s latest safety and efficacy data is that it will support the advancement of additional Cymerus™ MSC product candidates directly to Phase 2 trials in other indications. To this end, the company has begun planning for a Phase 2 trial in its next indication, critical limb ischemia (CLI).
The completion of this trial marks a major achievement for both Cynata and its partner for the GvHD field, Fujifilm. It is also a world-first for iPSC-derived therapeutic products, cementing Cynata’s position as a leader in new generation cell therapies.
Based on the results reported so far in the GvHD trial, Cynata is commercializing a safe and effective iPSC-derived cell therapeutic. This observation, together with compelling pre-clinical data in a range of disease models, provides a sound basis for the Company to consider future clinical trial possibilities for its proprietary Cymerus™ technology.
To learn more, view our interview with Dr. Ross Macdonald, CEO of Cynata Therapeutics.




















You may want to look at richsourcestemcells.com as another option.
This is incredible news! This stock is ready to soar!