|
|
Introduction
From β-islets for diabetes (1) to dopaminergic neurons (Figure 1) for Parkinson’s disease (2), induced pluripotent stem cell (iPSC)-derived, haplotyped, and multiplex-engineered allogeneic cell therapeutics empower the development of effective and safe Cell and Gene Therapy Products (CGTs) that could be manufactured on a large scale in a cost-effective manner and provide a functional cure for diseases that plague humankind today.
Chimeric antigen receptor iPSC-derived T (iCAR-T) and CAR natural killer (iCAR-NK) cell immunotherapies have already shown great promise in Phase I of several clinical trials by of hematological and other malignancies (3) demonstrating the vast potential of universal off-the-shelf iPSC-based cell and gene therapy to accelerate human health outcomes and reduce the socioeconomic burden of the world’s deadliest diseases.
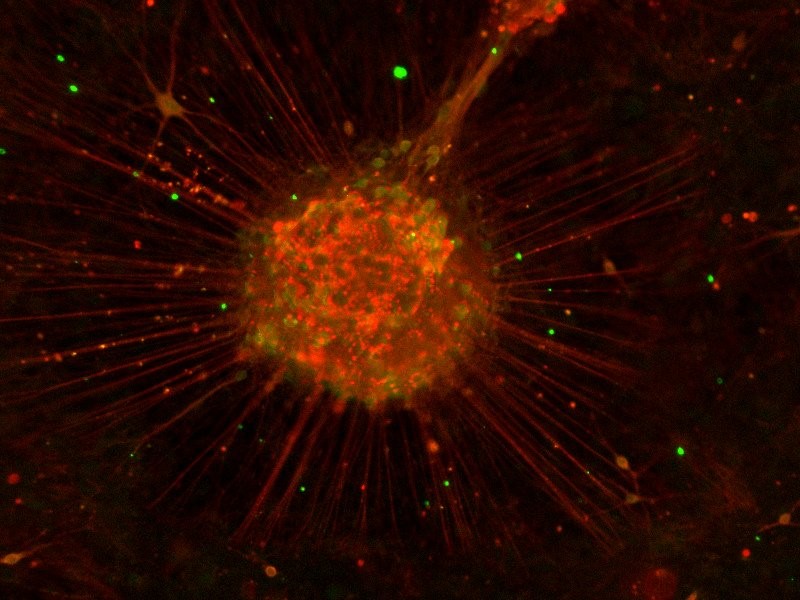
Figure 1. iPSC-derived dopaminergic neurons stained for Tuj1 (red) and Pitx3 (green).
To enable development of iPSC-derived cell therapeutics with scalable, cGMP-compliant manufacturing and banking processes, several biotech companies offer clinical iPSC generation services. Among stem cell pioneer biotech companies, REPROCELL has leveraged its long-standing expertise in mRNA-based reprogramming to offer a cGMP iPSC generated through its own mRNA-based, zero-footprint reprogramming technology.
However, with so many options from different providers who are offering cGMP-compliant iPSCs, it is difficult to know what to look for when choosing the right partner specialized in clinical-grade iPSCs and cell therapy manufacturing. What are the important factors to consider when evaluating clinical iPSC providers, so you do not run into complications later?
Is it the associated costs of generation and characterization of iPSCs, the reprogramming technology, the licensing terms, the starting material, or something else? To alleviate confusion, in this article, we discuss some of the main factors to consider when choosing the right partner to develop your cGMP-compliant iPSC Master Cell Bank (MCB).
Step 1: Regulatory compliance of donor consent and screening
Prior to any laboratory work, the basis for sourcing an eligible donor must be ensured. At the time of tissue collection, the donor must provide written informed consent and be screened to ensure the safety of the process and to protect the donor, lab workers, and future patients. This process must follow certain guidelines covering ethical compliance and donor eligibility including a medical history interview that details any exclusion or inclusion criteria that are key for your project.
One important consideration for these projects is whether the donor consented to participate in an iPSC-derived therapeutic project for commercial and clinical use. Depending on your project goals, additional language could state that the donor cells and their derivatives thereof will be used for genome engineering (e.g., CRISPR/Cas-9-based gene editing) or molecular screening such as genomic and transcriptomic analyses including NGS and RNA-sequencing.
The donor and the starting tissue must be authenticated and screened for the presence of pathogenic microorganisms. The panel of human pathogens to be screened for is specified by the regulatory agencies (e.g., US FDA, EU EMA, Japan PDMA). There are regional differences in the requirements of the virusespectrum that needs to be tested, the testing kits that must be used (certified by FDA or EMA) and the testing laboratories, which need to be certified in the US for FDA and EU accredited in an EU member state of the EMA.
In addition, some regulatory authorities demand re-testing of the donor blood in a second screen to accept eligible donor starting material prior to use in cell therapy. It is therefore important to choose a virus panel that meets the requirements of the regulatory body in the area where the therapy will ultimately be marketed.
Step 2: Source of Target Cells
Fibroblasts
One of the hurdles in developing an iPSC-based cell therapy program is the source of the target cells for reprogramming. Historically, most work with iPSCs has started with fibroblasts (Figure 2), which offer several advantages as a starting cell for reprogramming:
- Fibroblasts are readily isolated from a variety of sources, such as skin punch biopsies, foreskin, or surgical resections from plastic surgery.
- Primary fibroblasts are easy to culture and expand in vitro.
- The protocols for the reprogramming of primary fibroblasts using different non-integrating methods are fully optimized with a greater reprogramming efficiency compared to other primary cell types.
Some researchers are hesitant to use dermal fibroblasts as a starting material due to the chronic exposure of skin tissue to ultraviolet radiation (UVR) which is generally associated with a greater degree of DNA damage, thus leading to higher numbers of copy number variations (CNVs) (4). However, nowadays, the skin biopsies are usually taken from areas with minimal UV exposure.
Blood
More recently, iPSCs have been generated from other tissue sources, including blood-derived cells such as peripheral blood mononuclear cells (PBMCs) or umbilical cord blood mononuclear cells (CBMC). Blood-derived cells have the advantage that isolation of blood is generally less invasive for the donor than a skin biopsy, and PBMCs are a source of certain immune cells such as T- or B- cells which have undergone genomic rearrangement as part of their natural development. These genomic rearrangements will be retained in the subsequently generated iPSC clones derived from these cells (5). It is important to note that there is currently no factual evidence that blood-derived iPSCs differentiate more efficiently into various cells of the hematopoietic lineages e.g., NK cells compared to other starting cell types (6).
Other tissue sources
Other starting target cells, such as renal cells shed in urine, nasal scrapings, and dental pulp cells isolated from baby and adult teeth have been successfully reprogrammed with mRNA-based approach in a research setting. These tissue samples are attractive because their collection is less invasive for the donor than a blood draw or skin punch biopsy, and they provide potential sources of cells from pediatric donors. So far, these donor tissues have not been exploited much in a cGMP clinical setting but hold great promise for both autologous and heterologous iPSC-derived cellular therapies.
Step 3: Reprogramming method
There are three major reprogramming methods that have been used for generating iPSCs in a clinical setting.
- mRNA reprogramming provides the most reliable and safest non-integrating method for generating iPSCs. The mRNA reprogramming factors are not retained in the transfected cells longer than 48 to 72 hours due to natural breakdown of foreign RNAs in the cell (7) and therefore time-consuming screening for residual reprogramming vectors is redundant. In addition, mRNA reprogramming has the highest reprogramming efficiency rate among non-viral non-integrating methods (8) and results in the lowest rate of genomic abnormalities amongst the three methods (9). REPROCELL was the first company to develop commercial mRNA-based reprogramming in 2011, opening up an avenue of innovation and application of this effective technology to researchers around the world. REPROCELL has recently established the 4th Gen RNA Reprogramming Technology, which is used exclusively for clinical iPSC manufacturing service.
- Sendai virus vectors provide a viral vector-based method for expression of reprogramming factors in cells. These viral vectors contain viral RNAs that express transgenes without genomic integration that was a feature of prior viral methods (e.g., Retrovirus or Lentivirus). Sendai virus provides a reliable method for generating iPSCs. However, these virus reprogramming vectors can be retained for multiple passages, and therefore Sendai virus-derived iPSCs require labor intensive screening for residual reprogramming vectors (10).
- Episomal vectors, using DNA episomes to express the reprogramming transgenes, were used for some of the early clinical studies of iPSC-derived therapy. However, like Sendai-based methods, episomes can be retained in iPSCs for an extended period or could integrate foreign DNA into the genome (11), and episomal-derived iPSCs require extensive screening for retention of the vectors as well. In addition, episomal reprogramming requires specialized equipment for transfection (electroporation) that is not necessary for other methods. However, both Sendai virus and episomal vectors can reprogram blood suspension cells.
Step 4-6: Access to process intermediates (seed iPSCs)
In the iPSC MCB generation process described in Figure 3, the Sponsor can enter the workflow at several points, such as by providing starting material of suitable quality. For example, REPROCELL can facilitate this workflow through coordination with the client to ensure that their parental lines (i.e., fibroblasts) are compliant with the clinical standards and isolated from properly screened donors.
Additionally, REPROCELL offers clinically compatible seed bank clones, generated under strict quality assurance and control measures and are therefore suitable for further cGMP-compliant manufacturing into iPSC MCBs, either from our seed bank clone collection or from a custom iPSCs preparation made specifically for a client. Prior to generating a costly MCB, the client can determine first which clone is most suitable for their product development based on initial screening, genomic analyses, and phenotypic characterization.
Often researchers request to try different iPSC clones from different donors to assess donor clone parameters side-by-side using existing protocols. Screening multiple seed iPSC clones in parallel minimizes the risk and provides the clients with the assurance that the MCB generated from the selected clone will have the necessary performance during the downstream processing while keeping the initial costs low.
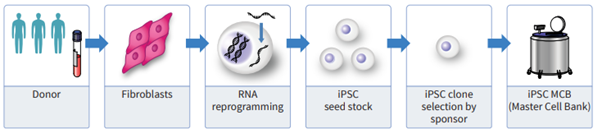
From left to right: The donor screened, the tissue is sourced, the target cells are isolated and reprogrammed to iPSCs as a clinical seed stock, clones are tested for suitability by the client, and a cGMP iPSC MCB is manufactured.
Other Considerations
The cGMP-compliant iPSC MCB manufacturing and the downstream processing to the final therapeutic medicinal product require different technical and quality assurance (QA) capabilities to meet the different regulatory requirements. Many CDMOs specialize in one aspect or another of the overall workflow (Figure 3), so collaborating with a service provider that has the capability, through a network of specialized partners, to supply clinical iPSCs and assist in the development of the final cell therapy product can simplify the process by:
- Providing overall project management.
- Facilitating material transfer from one process to the next.
- Overlapping MCB generation and therapeutic medicinal product manufacturing (such as technical transfer and process optimization).
Companies like REPROCELL have partnerships with several CDMOs that have cGMP-compliant manufacturing experiences of different cell-based clinical products, thereby providing the overall project management to ensure that the clients clinical cell therapy program runs to a successful conclusion.
Quality Assurance and Quality Control
The requirements for quality, traceability, and compliance of each step in the cGMP-compliant iPSC generation process is covered by guidelines and regulations from governmental agencies. Some regulations, such as the ICH Q5 regulations for biological products, are valid in multiple regions as they are harmonized international guidelines, while others such as the PMD Act in Japan are valid only in a subset of the regions. REPROCELL manufactures iPSCs by following the guidance from major health authorities (EMA, FDA, and PMDA), thus helping to ensure that your iPSCs are suitable for the global market.
Another element to consider is the additional requirements specific to a certain project, such as selection of specific genetic variants or genotypes. An iPSC service provider should work with a client to ensure that the donor eligibility/consent and quality control (QC) measures meet both their project requirements and the demands of the regulatory agencies. For example, REPROCELL encourages clients to audit the manufacturing process, including the QA/QC program of any third-party vendors that are used, to ensure that the provider’s programs meet the needed quality standards.
Licensing
Many researchers look only for the initial project costs but not for the hidden ones such as licensing fees. Most technologies used in any area of cell and gene therapy are covered by patents and must be licensed from the appropriate organizations. For reprogramming, some cGMP-compliant iPSC service providers use Sendai or episomal methods that are extensively covered by patents, and their use in a commercial clinical setting may require licenses from one or more organizations, each with their own royalty schemes, limitations on field of use, and uncertainties in final cost. Other providers, such as REPROCELL, have simplified commercialization terms to enable sponsors to obtain the necessary license with simple and predictable costs.
Each client should do their own due diligence regarding licensing since each project has different goals and uses different technologies for various targeted markets.
Conclusion
The generation of cGMP-compliant iPSCs is a pivotal step in developing of pluripotent stem cell-based therapeutics. There are several elements to consider when planning such complex project, including the source and compliance of the starting cell material, the selection of reprogramming method, QA and QC measures, and access to clinical iPSC intermediates for downstream process optimization.
In addition, consideration should be paid to the expertise of the chosen CDMO and to licensing the appropriate technology for reprogramming and the final therapeutic medicinal product. High-quality clinical iPSC service providers, such as REPROCELL, have full operating capability and broad expertise to guide you through the entire process of manufacturing a cell and gene therapy product.
1. Applications of iPSC-derived beta cells from patients with diabetes. Maxwell KG, Millman JR; Cell Rep Med. 2:100238 (2021)
2. Treatment of Parkinson’s disease through personalized medicine and induced pluripotent stem cells. Stoddard-Bennett T, Pera RR. Cells 8:26 (2019)
3. Engineered induced pluripotent stem cells for cancer immunotherapy. Zhou Y, Li M, et al. Cancers 14:2266 (2022).
4. Substantial somatic genomic variation and selection for BCOR mutations in human induced pluripotent stem cells. Rouhani FJ, Zou X, et al. Nature Genetics 54:1406 (2022)
5. The generate of human γδ T cell-derived induced pluripotent stem cells from whole peripheral blood mononuclear cell culture. Watanabe D, Koyanagi-Aoi M, et al. Stem Cells Translational Medicine 7:33 (2017).
6. iPSC Preparation and Epigenetic Memory: Does the Tissue Origin Matter? Scesa G, Adami R, Bottai D. Cells. 10:1470 (2021).
7. Efficient reprogramming of human fibroblasts and blood-derived endothelial progenitor cells using nonmodified RNA for reprogramming and immune evasion. Poleganov MA, Eminli S, et al. Human Gene Therapy 26:751 (2015)
8. RNA-based strategies for cell reprogramming toward pluripotency. Bailly A, Milhavet O, Lematre J-M. Pharmaceutics 14:317 (2022)
9. A comparison of non-integrating reprogramming methods. Schlaeger TM, Daheron L, et al. Nature Biotechnology 33:58 (2015)
10. Robust and highly efficient hiPSC generation from patient non-mobilized peripheral blood-derived CD34+ cells using the auto-erasable Sendai virus vector. Okamura T, Horie Y, et al. Stem Cell Res Ther 10, 185 (2019).
11. Umbilical Cord Tissue as a Source of Young Cells for the Derivation of Induced Pluripotent Stem Cells Using Non-Integrating Episomal Vectors and Feeder-Free Conditions. Mohamed A, Chow T, et al. Cells 10:49 (2020)


Tell Us What You Think!