Currently, the cell and gene therapy (CGT) field is on track for a serious capacity shortage. In particular, there is a significant shortage of manufacturing capacity at the commercial scale. Although nearly 90% of cell and gene therapy developers would prefer to use contract manufacturing organizations (CMOs), relevant CMO capacity is not yet available.
Moreover, the lead time for CMOs to begin cell and gene therapy projects now averages well over a year and sometimes as long as two. Therefore, CGT developers are often forced to expand their in-house capacity.
Contract Manufacturing for Cell and Gene Therapies
With the future in mind, contract manufacturing organizations (CMOs) and contract development and manufacturing organizations (CDMOs) are investing heavily in expanding their manufacturing capacity to be able to take on new clients and larger, later-stage projects as the market expands.
With several cell and gene therapy (CGT) companies actively progressing products through clinical trials toward commercialization, manufacturing capacity for CGT is becoming tight. Thus, we are seeing companies adjusting to this dynamic by signing supply agreements with CDMOs, building internal manufacturing capabilities, and improving manufacturing processes and efficiency by exploring new technologies.
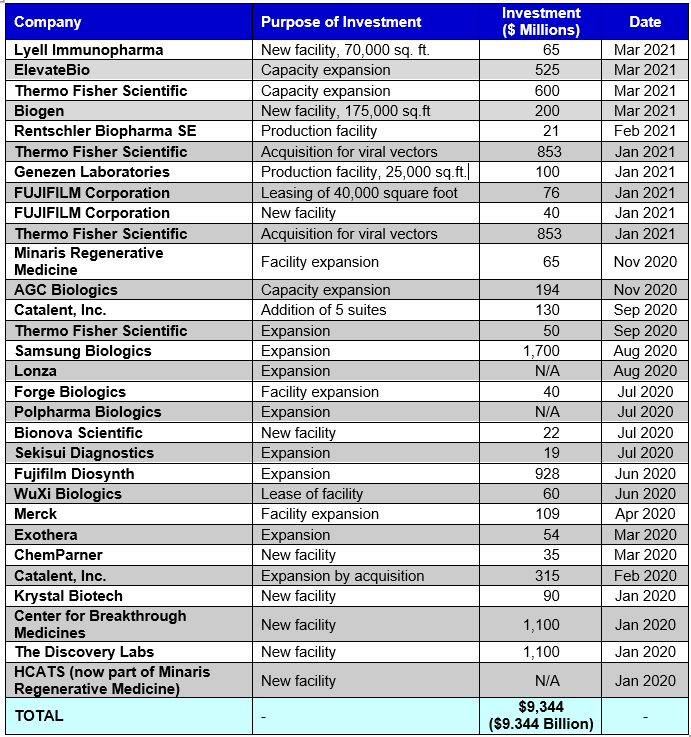
A roundup of recently announced facility expansions to support CGT manufacturing is provided in the table below. From January 2020 to present, nearly $10 billion has been invested into CMO/CDMO expansions to support cell and gene therapy manufacturing and the trend is continuing.
This incredible figure proves to investors and executives alike that this market is booming and permanently here to stay.
TABLE 1.1: Recent CMO/CDMO Expansions within the Cell and Gene Therapy Sector





















Tell Us What You Think!