MINNEAPOLIS, Dec. 9, 2021 — BlueCube Bio officially announced a company name transition to Evia Bio™, concurrent with the launch of a significant new cryopreservation technology for use in cell and gene therapies utilizing Induced Pluripotent Stem Cells (iPSCs). The osmolyte-based solution increases cell survival above that of commercially available DMSO-based options which is critical for realizing the potential of these therapies. This latest addition expands the company’s portfolio of patented cryopreservation products that also include products for mesenchymal stem cells (MSCs). [Read more…]
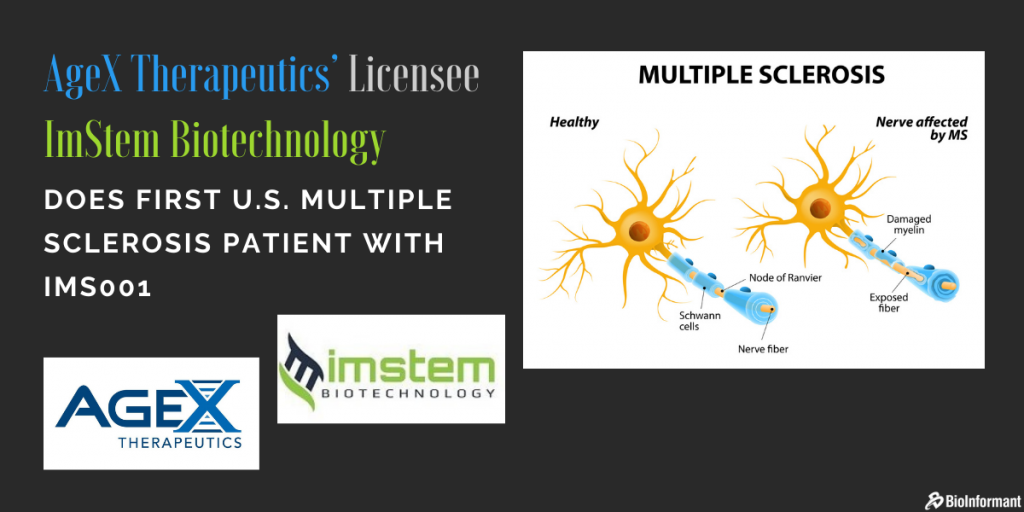
AgeX Therapeutics’ Licensee ImStem Biotechnology Does First U.S. Multiple Sclerosis Patient with IMS001
-
ImStem’s MSC Product Candidate IMS001 is Derived from AgeX’s Pluripotent Stem Cell Line ESI-053.
-
IMS001 is an investigational, allogeneic, MSC product derived from a human embryonic stem cell line.
-
It is assumed to be the first hES-MSC based allogeneic cell therapy accepted for clinical trial by the FDA for patients with MS.
denovoMATRIX releases isoMATRIX: Cell and Gene Therapy’s first high-yield MSC isolation technology
DRESDEN, Germany, December 15, 2021 / BioInformant / — denovoMATRIX GmbH, an innovator in cell and gene therapy (CGT) manufacturing, announced the launch of a new technology for bringing down costs and increasing access to high-quality mesenchymal stromal cells (MSCs) for stem cell-based therapies. The technology termed isoMATRIX, is designed to isolate MSCs in xeno-/serum-free conditions with high efficiency and stemness enabling rapid cell expansion for radically reduced cost of goods (COGs) for cell therapy manufacturers.
Winner of the European EIC Accelerator program, EVerZom accelerates the industrialization of its exosome bioproduction platform for regenerative medicine
Paris, 18 November 2021 — EVerZom, a biopharmaceutical company specialized in the biomanufacturing of extracellular vesicles (or exosomes), announced today that it has obtained non-dilutive funding of €2.5 million as the winner of the European EIC Accelerator competition (Horizon Europe). This funding will enable the company to transfer its extracellular vesicle biomanufacturing technology for a first clinical grade batch production and to accelerate the scale-up of its production capabilities by developing a process compatible with clinical Phase 2/3 for the pharmaceutical industry. [Read more…]
Saint-Gobain Life Sciences and denovoMATRIX Announce Collaboration to Optimize New Product Development
DRESDEN, Germany, 9 November, 2021 / BioInformant / — denovoMATRIX GmbH and Saint-Gobain Life Sciences have entered into a Master Service Agreement focused on optimizing the performance of new products in development for adherent cell culture and harvest for autologous therapies. The relationship will further explore the synergy of consumable products from both parties and their positive effects for the therapeutic market. [Read more…]
- « Previous Page
- 1
- …
- 63
- 64
- 65
- 66
- 67
- …
- 116
- Next Page »