Proudly Supporting Great Science and Women in STEM
WORD+ 2025 is pleased to introduce its conference chairs and speakers.
FYI: In February 2025, 60% of session chairs and 54% of speakers will be leading scientists who are female. [Read more…]
Cellistic® Announces Successful Certification of its GMP Facility Dedicated to iPSC-Based Off-the-shelf Cell Therapy Manufacturing Solutions
MONT-SAINT-GUIBERT, Belgium — Cellistic®, a pioneer in iPSC-based off-the-shelf cell therapy development and manufacturing, announces the successful audit and GMP certification of its first-of-its kind facility dedicated to manufacturing therapies based on induced pluripotent stem cells (iPSCs). This achievement signifies Cellistic’s quality and regulatory readiness, which is now certified by the Belgian Federal Agency for Medicines and Health Products (FAMHP), operating under European Medicines Agency (EMA) guidelines. [Read more…]
Autolus Latest Competitor in Global CAR-T Frenzy with FDA Approval of Aucatzyl
On November 8, 2024, the U.S. FDA issued a historic approval of the seventh CAR-T cell therapy to be approved in the U.S. and the 11th CAR-T therapy to be approved globally. This approval was for Autolus Inc.’s Aucatzyl (obecabtagene autoleucel), a CD19-directed genetically modified autologous T cell immunotherapy, for adults with relapsed or refractory B-cell precursor acute lymphoblastic leukemia (ALL). Autolus is a U.K. biotech company that develops novel gene-modified T cell therapies for the treatment of cancer. [Read more…]
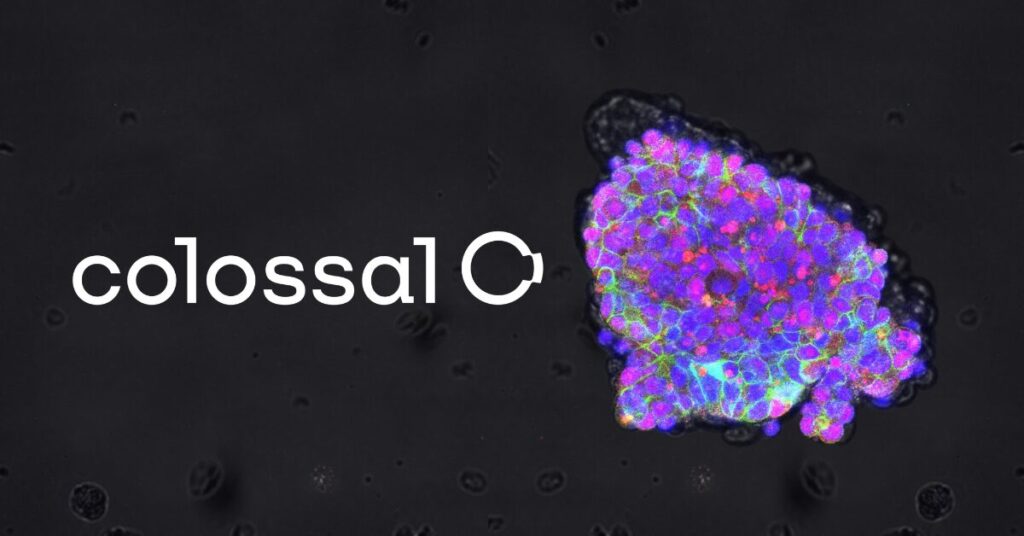
Colossal Achieves Multiple Scientific Firsts in Progress Towards Thylacine De-Extinction
Successes across the de-extinction pipeline signal that the thylacine project is on track as Colossal continues its global effort to fight the extinction crisis.
Oct 16, 2024, DALLAS & MELBOURNE, Australia — Colossal, the de-extinction and species preservation company, announces numerous breakthrough successes in all stages of the thylacine de-extinction effort that put the company much closer to returning the iconic thylacine to Australia. From the generation of the highest quality ancient genome to date, to breaking records in multiplex genome editing, to innovations in assisted reproductive technologies for marsupials, Colossal’s success in developing de-extinction technologies will not just support the important thylacine effort but will also help combat the world’s extinction crisis. [Read more…]
- « Previous Page
- 1
- …
- 18
- 19
- 20
- 21
- 22
- …
- 116
- Next Page »