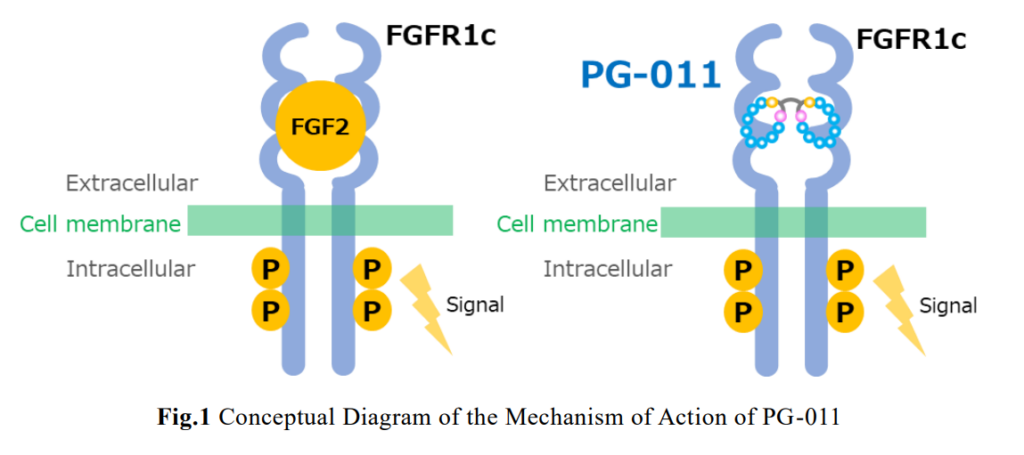
PeptiGrowth Inc. (Headquarters: Chiyoda-ku, Tokyo; President: Jiro Sugimoto) has successfully developed a novel synthetic peptide called “FGF2 alternative peptide (FGFR1c agonist)”, which has equivalent function to recombinant FGF2 (Fibroblast Growth Factor 2, basic FGF (bFGF)). This product will be on the market at the beginning of October 2024. [Read more…]
Cell X Technologies and Aspen Neuroscience collaborate to address throughput and scalability in manufacturing automation to facilitate iPSC cell processing
Advancing the promise of cell-replacement therapy for people with Parkinson’s disease
CLEVELAND, Ohio / SAN DIEGO, Calif. – October 8, 2024 – Cell X Technologies, pioneers in automated cell processing solutions, and Aspen Neuroscience, Inc., dedicated to autologous regenerative medicine, have entered into a collaboration and licensing agreement focused on providing an automated solution for Aspen’s iPSC-derived autologous clinical manufacturing. [Read more…]
Immunofluorescent Staining Within Hours, Not Days
AMSBIO has launched CellO-IF – an all-in-one immunofluorescent staining reagent kit designed to accelerate the labelling organoids and spheroids directly in hydrogels or extracellular matrices, while preserving delicate structures and cellular integrity.
Immunofluorescence (IF) allows detection and localization of antigens in diverse types of tissues in various cell preparations. The technique provides excellent sensitivity and amplification of signal by comparison to immunohistochemistry. However, traditional IF staining is a labor intensive, multi-step process. CellO-IF revolutionizes this by streamlining the procedure. [Read more…]
South Korea’s SK bioscience Completes Acquisition of Germany’s CDMO IDT Biologika
SK bioscience completed the acquisition of German CDMO company IDT Biologika with over a century of history that enables immediate expansion in revenue and infrastructure.
The company aims to double sales of IDT Biologika by 2028 through new business projects and operational efficiency.
The company will invest in high-growth businesses such as stage cell and gene therapy (CGT) and recombinant vaccine.
SK bioscience will accelerate its global push based on the complementary capabilities of both companies.
RxCell Inc. Granted U.S. Patent for iPSC-based Cell Therapy
PARK CITY, Utah, September 19, 2024 – RxCell Inc. is pleased to announce the issuance of U.S. Patent 11,946,069 entitled “Method for Generating Multiple Cellular Products from Single Pluripotent Cell Source”, a significant advancement in the field of regenerative medicine. This patent, granted on April 2, 2024, provides RxCell with exclusive rights to induced pluripotent stem cell (iPSC)-derived cells for therapy across a broad spectrum of diseases. [Read more…]
- « Previous Page
- 1
- …
- 20
- 21
- 22
- 23
- 24
- …
- 116
- Next Page »