Passed December 13, 2016, the 21st Century Cures Act authorizes $6.3 billion in funding. It will support the development of innovative regenerative medicines, by providing substantial funding to the U.S. NIH and the FDA, among other groups.
21st Century Cures Act
In this article:
- What Is the 21st Century Cures Act?
- The 21st Century Cures Act Summary
- 21st Century Cures Act FDA | Cancer Research
- 21st Century Cures Act Funding
- 21st Century Cures Act Controversy
- Learn More About 21st Century Cures Act
What Is the 21st Century Cures Act?
The 21st Century Cures Act was passed by both houses of Congress and signed into law by President Obama. It puts the U.S. on the same playing field as other regions, such as Japan, South Korea, and the EU, which have accelerated regulatory pathways for regenerative therapies.
The Role of Regenerative Medicine
The bill encourages late-stage development in regenerative medicine, recognizing the field’s potential to address severe unmet medical needs. It also correctly identifies regenerative therapy as a category in its own right.
The initiative will also support President Obama’s cancer “moonshot,” Alzheimer’s research, and efforts to address opioid addiction.
The 21st Century Cures Act Summary
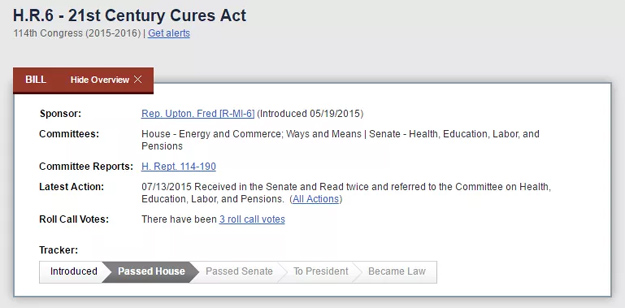
According to the U.S. FDA, the 21st Century Cures Act “builds on FDA’s ongoing work to incorporate the perspectives of patients into the development of drugs, biological products, and devices in FDA’s decision-making process.”
The purpose of the 21st Century Cures Act is clear: To accelerate the discovery, development, and delivery of innovative cures.
21st Century Cures Act FDA | Cancer Research
On December 13, 2016, President Obama signed the 21st Century Cures Act into law, authorizing $6.3 billion in funding. In an emotional signing that was one of the last of his presidency, Obama stated, “Let’s make America the country to solve cancer.”
Approval of the Cure Act
Previously, the bill was approved 392-26 by the House of Representatives and 94-5 in the U.S. Senate.
The bill authorizes large funding amounts for both the U.S. NIH and the U.S. FDA.
21st Century Cures Act Funding
As part of the bill, substantial support is provided to the NIH, including the establishment of an NIH and Cures Innovation Fund, which will appropriate funds:
(1) for biomedical research, including high-risk, high-reward research and research conducted by early stage investigators;
(2) to develop and implement a strategic plan for biomedical research; and
(3) to carry out specified provisions of the Act.
The act will also authorize up to $500M for the U.S. FDA to assist with regulatory support.
21st Century Cures Act Controversy
Concerns over the bill surround the involvement of private industry, with 400+ companies lobbying for it, including 58 pharmaceutical companies, 26 biotechnology companies, and 24 medical device companies. (Data courtesy of Shelly Ross, Cure Alliance.)
The Opposition
A rare opponent to the bill, Senator Bernie Sanders suggested that it does not do enough to “curtail” the high costs of prescription drugs.
Advancing U.S. Healthcare
Nonetheless, the 21st Century Cures Act is a major step forward, and one that is largely supported by all but the most conservative members of the scientific and medical communities. To read the 21st Century Cures Act full text, click here.
Learn More About 21st Century Cures Act
Proposed FDA oversight of the regenerative medicine field should ensure high standards of quality and safety, facilitate early patient access to therapies with proven efficacy, and address serious and life-threatening conditions.
To learn more about the law, watch the video below:
If you found this article valuable, subscribe to BioInformant’s stem cell industry updates.
We are the industry leaders in stem cell research, with research cited by The Wall Street Journal, Xconomy, AABB, and Vogue Magazine. Bringing you breaking news on an ongoing basis, join more than half a million loyal readers, including physicians, scientists, investors, and philanthropists.
Have thoughts, perspectives, or opinions on the 21st Century Cures Act? Share them in the comments section below.
Up Next: What is an RMAT Designation and Who Has One?
Editor’s Note: This post was originally published on March 13, 2018, and has been updated for quality and relevancy.




















No law is perfect, but as a physician I’m excited personally and professionally to see this legislation move forward. It’s time to let the U.S. use its creative and intellectual capital to move ahead and catch up to other countries who have already recognized the healing potential of regenerative medicine and made it a priority. Harnessing the body’s potential to heal itself is unquestionably the #futureofmedicine. Sincerely, Alan J. Bauman, M.D.
It wasn’t the REGROW Act, it was more in line with what the FDA wanted in order to align with Europe and Japan. The specifics will be worked out over some years, but in the meantime it should accelerate approvals of “drugs” already in clinical trials. Also the link to the Act is not to the right Act, its a bit tricky to get to, but I finally got there today. This is to the enrolled text, approved by both houses.
https://www.congress.gov/bill/114th-congress/house-bill/34/text